|
TEACHING
OBJECTIVES
Understand
the concept and significance of tolerance
Know
the factors which determine induction of tolerance
Understand
the mechanism of tolerance induction
Understand
the concepts of autoimmunity and disease
Know
the features of major autoimmune diseases
Know
the theories on etiology of autoimmune disease |
TOLERANCE
Introduction
Tolerance refers to the specific
immunological non-reactivity to an antigen resulting from a previous exposure to
the same antigen. While the most important form of tolerance is non-reactivity
to self antigens, it is possible to induce tolerance to non-self antigens. When
an antigen induces tolerance, it is termed tolerogen.
Tolerance to self antigens
We normally do not mount a
strong immune response against our own (self) antigens, a phenomenon
called self-tolerance. When the immune system recognizes a self antigen
and mounts a strong response against it, autoimmune disease develops.
Nonetheless, the immune system has to recognize self-MHC to mount a
response against a foreign antigen. Thus, the immune system is
constantly challenged to discriminate self vs non-self and mediate the
right response.
Induction of tolerance to non-self
Tolerance can also be induced to
non-self (foreign) antigens by modifying the antigen, by injecting the antigen
through specific routes such as oral, administering the antigen when the immune
system is developing, etc. Certain bacteria and viruses have devised clever ways
to induce tolerance so that the host does not kill these microbes. Ex: Patients
with lepromatous type of leprosy do not mount an immune response against Mycobacterium leprae.
Tolerance to tissues and cells
Tolerance to tissue and cell
antigens can be induced by injection of hemopoietic (stem) cells in neonatal or
severely immunocompromised (by lethal irradiation or drug treatment) animals.
Also, grafting of allogeneic bone marrow or thymus in early life results in tolerance to the donor type
cells and tissues. Such animals are known as chimeras. These findings are
of significant practical application in bone marrow grafting.
|
| |
Tolerance to soluble antigens
A state of tolerance to a variety
of T-dependent and T-independent antigens has been achieved in various
experimental models. Based on these observations it is clear that a number of
factors determine whether an antigen will stimulate an immune response or
tolerance (Table 1).
|
Table 1
Factors
that determine induction of immune response or tolerance following
challenge with antigen |
|
Factors that affect
response to Ag |
Favor immune response |
Favor tolerance |
|
Physical form of
antigen |
Large, aggregated,
complex molecules; |
Soluble,
aggregate-free, relatively smaller, less complex molecules, Ag not
processed by APC or processed by cell without class II MHC |
|
Route of Ag
administration |
Sub-cutaneous or
intramuscular |
Oral or sometimes
intravenous |
|
Dose of antigen |
Optimal dose |
Very large (or sometime
very small) dose |
|
Age of responding
animal |
Older and
immunologically mature |
Newborn (mice),
immunologically immature |
|
Differentiation state
of cells |
Fully differentiated
cells; memory T and memory B cells |
Relatively
undifferentiated: B cells with only IgM (no IgD), T cells (e.g.
cells in thymic cortex) |
Immunologic features of tolerance
Tolerance is different from
non-specific immunosuppression and immunodeficiency. It is an active antigen-dependent process in response to the antigen. Like immune response, tolerance is
specific and like immunological memory, it can exist in T-cells, B cells or both
and like immunological memory, tolerance at the T cell level is longer lasting
than tolerance at the B cell level.
Induction of tolerance in T cells
is easier and requires relatively smaller amounts of tolerogen than tolerance in
B cells. Maintenance of immunological tolerance requires persistence of antigen.
Tolerance can be broken naturally (as in autoimmune diseases) or artificially
(as shown in experimental animals, by x-irradiation, certain drug treatments and
by exposure to cross reactive antigens).
Tolerance may be induced to all
epitopes or only some epitopes on an antigen and tolerance to a single antigen
may exist at the B cell level or T cell level or at both levels.
|
| |
Mechanism of tolerance induction
The exact mechanism of induction
and maintenance of tolerance is not fully understood. Experimental data,
however, point to several possibilities.
Clonal deletion
T and B lymphocytes during
development come across self antigens and such cells undergo clonal
deletion through a process known as apoptosis or programmed cell death.
For example, T cells that develop in the thymus first express neither
CD4 nor CD8. Such cells next acquire both CD4 and CD8 called
double-positive cells and express low levels of
αβ TCR. Such cells
undergo positive selection after interacting with class I or class II
MHC molecules expressed on cortical epithelium. During this process,
cells with low affinity for MHC are positively selected. Unselected
cells die by apoptosis, a process called "death by neglect". Next, the
cells loose either CD4 or CD8. Such T cells then encounter self-peptides
presented by self MHC molecules expressed on dendritic cells. Those T
cells with high affinity receptors for MHC + self-peptide undergo clonal
deletion also called negative selection through induction of apoptosis.
Any disturbance in this process can lead to escape of auto-reactive
T-cells that can trigger autoimmune disease. Likewise, differentiating
early B cells when they encounter self-antigen, cell associated or
soluble, undergo deletion. Thus, clonal deletion plays a key role in
ensuring tolerance to self antigen.
Peripheral tolerance
The clonal deletion is not a fool proof system and
often T and B cells fail to undergo deletion and therefore such cells
can potentially cause autoimmune disease once they reach the peripheral
lymphoid organs. Thus, the immune system has devised several additional
check points so that tolerance can be maintained.
Activation-induced cell death
T cells upon activation not only produce
cytokines or carryout their effector functions but also die through
programmed cell death or apoptosis. In this process, the death receptor
(Fas) and its ligand (FasL) play a crucial role. Thus, normal T cells
express Fas but not FasL. Upon activation, T cells express FasL which
binds to Fas and triggers apoptosis by activation of caspase-8. The
importance of Fas and FasL is clearly demonstrated by the observation
that mice with mutations in Fas (lpr mutation) or FasL (gld mutation)
develop severe lymphoproliferative and autoimmune disease and die within
6 months while normal mice live up to 2 years. Similar mutations in
these apoptotic genes in humans leads to a lymphoproliferative disease
called autoimmune lymphoproliferative syndrome (ALPS).
Clonal anergy
Auto-reactive T cells
when exposed to antigenic peptides on antigen presenting cells (APC)
that do not possess the co-stimulatory molecules CD80 (B7-1) or CD86
(B7-2) become anergic (nonresponsive) to the antigen. Also, while
activation of T cells through CD28 triggers IL-2 production, activation
of CTLA4 leads to inhibition of IL-2 production and anergy. Also, B
cells when exposed to large amounts of soluble antigen down-regulate
their surface IgM and become anergic. These cells also up-regulate the
Fas molecules on their surface. An interaction of these B cells with
Fas-ligand bearing T cells results in their death via apoptosis.
Clonal ignorance
T cells reactive to
self-antigen not represented in the thymus will mature and migrate to
the periphery, but they may never encounter the appropriate antigen
because it is sequestered in inaccessible tissues. Such cells may die
out for lack of stimulus. Auto-reactive B cells, that escape deletion,
may not find the antigen or the specific T-cell help and thus not be
activated and die out.
Anti-idiotype antibody
These are antibodies that are
produced against the specific idiotypes of other antibodies. Anti-idiotypic
antibodies are produced during the process of tolerization and have been
demonstrated in tolerant animals. These antibodies may prevent the B
cell receptor from interacting with the antigen.
Regulatory T cells (Formerly called suppressor cells)
Recently, a distinct
population of T cells has been discovered called regulatory T cells.
Regulatory T cells come in many flavors, but the most well characterized
include those that express CD4+ and CD25+. Because activated normal CD4
T cells also express CD25, it was difficult to distinguish regulatory T
cells and activated T cells. The latest research suggests that
regulatory T cells are defined by expression of the forkhead family
transcription factor Foxp3. Expression of Foxp3 is required for
regulatory T cell development and function. The precise mechanism/s
through which regulatory T cells suppress other T cell function is not
clear. One of the mechanisms include the production of immunosuppressive
cytokines such as TGF-β and IL-10. Genetic mutations in Foxp3 in humans
leads to development of a severe and rapidly fatal autoimmune disorder
known as Immune dysregulation, Polyendocrinopathy, Enteropathy, X-linked
(IPEX) syndrome. This disease provides the most striking evidence that
regulatory T cells play a critical role in preventing autoimmune
disease.
Termination of tolerance
Experimentally induced tolerance
can be terminated by prolonged absence of exposure to the tolerogen, by
treatments which severely damage the immune system (x-irradiation) or by
immunization with cross reactive antigens. These observations are of
significance in the conceptualization of autoimmune diseases.
|
| |
AUTOIMMUNITY
Definition
Autoimmunity can be defined as
breakdown of mechanisms responsible for self tolerance and induction of an
immune response against components of the self. Such an immune response may not
always be harmful (e.g., anti-idiotype antibodies). However, in numerous
autoimmune diseases it is well recognized that products of the immune system
cause damage to the self.
Effector mechanisms in autoimmune
diseases
Both antibodies and effector T
cells can be involved in the damage in autoimmune diseases.
General classification
Autoimmune diseases are generally
classified on the basis of the organ or tissue involved. These diseases may fall
in an organ-specific category in which the immune response is directed against
antigen(s) associated with the target organ being damaged or a
non-organ-specific category in which the antibody is directed against an antigen
not associated with the target organ (Table 2). The antigen involved in most
autoimmune diseases is evident from the name of the disease (Table 2).
Genetic predisposition for
autoimmunity
Studies in mice and observations in
humans suggest a genetic predisposition for autoimmune diseases. Association
between certain HLA types and autoimmune diseases has been noted (HLA: B8, B27,
DR2, DR3, DR4, DR5 etc.).
|
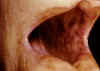 Figure 1 Hyperpigmentation of buccal mucosa in Addison's disease © Bristol Biomedical
Archive. Used with permission
Figure 1 Hyperpigmentation of buccal mucosa in Addison's disease © Bristol Biomedical
Archive. Used with permission
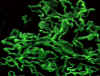 Figure 2 Immunofluorescent stain of immunoglobulin G (IgG) showing linear pattern in Goodpasture's
Figure 2 Immunofluorescent stain of immunoglobulin G (IgG) showing linear pattern in Goodpasture's
syndrome ©
Bristol Biomedical Archive. Used with permission
 Figure 3 Pemphigus vulgaris - immunofluorescence
©
Bristol Biomedical Archive. Used with permission
Figure 3 Pemphigus vulgaris - immunofluorescence
©
Bristol Biomedical Archive. Used with permission
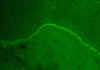 Figure 4 Mucous membrane pemphigoid - immunofluorescence
© Bristol Biomedical Archive.
Used with permission
Figure 4 Mucous membrane pemphigoid - immunofluorescence
© Bristol Biomedical Archive.
Used with permission
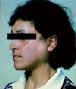 Figure 5 Parotid enlargement in Sjogren's syndrome ©
Bristol Biomedical Archive. Used with permission
Figure 5 Parotid enlargement in Sjogren's syndrome ©
Bristol Biomedical Archive. Used with permission
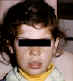
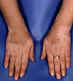 Figure 6 Depigmentation in vitiligo ©
Bristol Biomedical Archive. Used with permission
Figure 6 Depigmentation in vitiligo ©
Bristol Biomedical Archive. Used with permission
|
Etiology of autoimmunity disease
The exact etiology of autoimmune
diseases is not known. However, various theories have been offered. These
include sequestered antigen, escape of auto-reactive clones, loss of suppressor
cells, cross reactive antigens including exogenous antigens (pathogens) and
altered self antigens (chemical and viral infections).
Sequestered antigen
Lymphoid cells may not be exposed to some self antigens during their
differentiation, because they may be late-developing antigens or may be confined
to specialized organs (e.g., testes, brain, eye, etc.). A release
of antigens from these organs resulting from accidental traumatic injury or
surgery can result in the stimulation of an immune response and initiation of an
autoimmune disease.
Escape of auto-reactive clones
The negative selection in the thymus may not be fully functional to eliminate
self reactive cells. Not all self antigens may be represented in the thymus or
certain antigens may not be properly processed and presented.
Lack of regulatory T cells
There are fewer regulatory T-cells in many autoimmune diseases.
|
Table 2
Spectrum of
autoimmune diseases, target organs and diagnostic tests |
|
|
Disease |
Organ |
Antibody to |
Diagnostic Test |
| Organ-Specific

Non-organ Specific
|
Hashimoto's thyroiditis
|
Thyroid |
Thyroglobulin,
thyroid peroxidase (microsomal) |
RIA, CF, hemagglutination
|
|
Primary Myxedema |
Thyroid |
Cytoplasmic TSH receptor
|
Immunofluorescence
(IF) |
| Graves'
disease |
Thyroid |
|
Bioassay, Competition for TSH receptor
|
| Pernicious
anemia |
Red cells |
Intrinsic
factor (IF), Gastric parietal cell |
B-12
binding to IF immunofluorescence
|
Addison's
disease
(Fig 1) |
Adrenal |
Adrenal
cells |
Immunofluorescence |
|
Premature onset menopause
|
Ovary |
Steroid
producing cells |
Immunofluorescence |
|
Male infertility |
Sperm |
Spermatozoa |
Agglutination, Immunofluorescence
|
|
Insulin dependent juvenile diabetes
|
Pancreas |
Pancreatic
islet beta cells |
|
|
Insulin resistant diabetic
|
Systemic |
Insulin
receptor |
Competition for
receptor |
| Atopic
allergy |
Systemic |
beta-adrenergic receptor
|
Competition for
receptor |
| Myasthenia graves |
Muscle |
Muscle, acetyl choline receptor
|
Immunofluorescence, competition for
receptor
|
|
Goodpasture's syndrome
|
Kidney, lung
|
Renal and lung basement
membrane |
Immunofluorescence
(linear staining) (Fig. 2) |
| Pemphigus |
Skin |
Desmosomes |
Immunofluorescence
(Fig 3) |
| Pemphigoid |
Skin |
Skin basement membrane
|
Immunofluorescence
(Fig 4) |
| Phacogenic uveitis |
Lens |
Lens protein |
|
| AI hemolytic anemia |
Erythrocytes
Platelet |
Erythrocytes |
Passive
hemagglutination
Direct Coomb's
test |
|
Idiopathic thrombocytopenia
|
|
Platelet |
Immunofluorescence |
|
Primary biliary cirrhosis
|
Liver |
Mitochondria |
Immunofluorescence |
|
Idiopathic neutropenia
|
Neutrophils
|
Neutrophils |
Immunofluorescence |
| Ulcerative colitis |
Colon |
Colon lipopolysaccharide
|
Immunofluorescence |
| Sjogren's
syndrome |
Secretory glands
(Fig 5)
|
Duct mitochondria |
Immunofluorescence |
| Vitiligo |
Skin Joints
|
Melanocytes
(fig 6) |
Immunofluorescence |
| Rheumatoid
arthritis |
Skin, kidney,
joints etc |
IgG |
IgG-latex agglutination |
|
Systemic lupus
erythematosus
|
joints, etc.
|
DNA, RNA,
nucleoproteins
|
RNA-, DNA-latex
agglutination, IF
(granular in kidney)
|
|
|
|
|
|
|
Diseases are listed from the most organ-specific (top) to the least specific
(bottom) |
Cross reactive antigens
Antigens on certain pathogens may have determinants which cross react with self antigens and an immune response against these determinants may lead to
effector cell or antibodies against tissue antigens. Post streptococcal
nephritis and carditis, anticardiolipin antibodies during syphilis and
association between Klebsiella and ankylosing spondylitis are examples of
such cross reactivity.
Diagnosis
Diagnosis of autoimmune diseases is
based on symptoms and detection of antibodies (and/or very early T cells) reactive against
antigens of tissues and cells involved. Antibodies against cell/tissue
associated antigens are detected by immunofluorescence. Antibodies against
soluble antigens are normally detected ELISA or radioimmunoassay (see table
above). In some cases, a biological /biochemical assay may be used (e.g.,
Graves diseases, pernicious anemia).
Treatment
The goals of treatment of autoimmune disorders are to
reduce symptoms and control the autoimmune response while maintaining the body's
ability to fight infections. Treatments vary widely and depend on the specific
disease and symptoms: Anti-inflammatory (corticosteroid) and immunosuppressive
drug therapy (such as cyclophosphamide, azathioprine, cyclosporine ) is the
present method of treating autoimmune diseases. Extensive research is being
carried out to develop innovative treatments which include: anti-TNF alpha
therapy against arthritis, feeding antigen orally to trigger tolerance, anti-idiotype
antibodies, antigen peptides, anti-IL2 receptor antibodies, anti-CD4 antibodies,
anti-TCR antibodies, etc.
Models of autoimmune diseases
There are a number of experimental
and natural animal models for the study of autoimmune diseases. The experimental
models include experimental auto-allergic encephalitis, experimental
thyroiditis, adjuvant induced arthritis, etc.
Naturally occurring models of
autoimmune diseases include hemolytic anemia in NZB mice,
systemic lupus erythematosus in NZB/NZW (BW), BXSB and MRL
mice and diabetes in obese mice.
|

 Figure 1 Hyperpigmentation of buccal mucosa in Addison's disease © Bristol Biomedical
Archive. Used with permission
Figure 1 Hyperpigmentation of buccal mucosa in Addison's disease © Bristol Biomedical
Archive. Used with permission






