| X | X | ||||
 |
|||||
| INFECTIOUS DISEASE | BACTERIOLOGY | IMMUNOLOGY | MYCOLOGY | PARASITOLOGY | VIROLOGY |
|
|||||
| TURKISH | |||||
| ALBANIAN | |||||
|
Let us know what you think FEEDBACK |
|||||
| SEARCH | |||||
|
|
|||||
|
|
|||||
 Figure 1
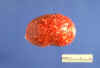
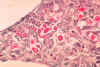
Figure 1Brewer's yeast (also known as Baker's yeast) with bud and bud scars (Saccharomyces cerevisiae). © Dennis Kunkel Microscopy, Inc. Used with permission |
Yeasts are single-celled budding organisms (figure 1). They do not
produce mycelia. The colonies are usually visible on the plates in 24 to
48 hours. Their soft, moist colonies resemble bacterial cultures rather
than molds. There are many species of yeasts that can be pathogenic for
humans. We shall only discuss the three most
significant species:
|
||||
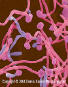 Figure 2
Figure 2Candida albicans - yeast and hyphae stages. A yeast-like fungus commonly occuring on human skin, in the upper respiratory, alimentary & female genital tracts. This fungus has a dimorphic life cycle with yeast and hyphal stages. The yeast produces hyphae (strands) and pseudohyphae. The pseudohyphae can give rise to yeast cells by apical or lateral budding. Causes candidiasis which includes thrush (an infection of the mouth and vagina) and vulvo-vaginitis. © Dennis Kunkel Microscopy, Inc. Used with permission |
CANDIDIASIS (Candida
albicans)
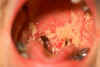
There are more than 20 species of the genus Candida that cause disease. The infections caused by all species of Candida are called candidiasis. Candida albicans (figure 2 and 3) is an endogenous organism and the most common species in human infections. It can be found in 40 to 80% of normal human beings. It is present in the mouth (figure 4), gut, and vagina. It may be present as a commensal or a pathogenic organism.
Infections with Candida usually occur when a patient has some alteration in cellular immunity, normal flora or normal physiology. Patients with decreased cellular immunity have decreased resistance to fungal infections. Prolonged antibiotic or steroid therapy destroys the balance of normal flora in the intestine allowing the endogenous Candida to overcome the host. Invasive procedures, such as cardiac surgery and indwelling catheters, produce alterations in host physiology and some of these patients develop Candida infections. Although it most frequently infects the skin and mucosae, Candida can cause pneumonia, septicemia or endocarditis in the immuno-compromised patient. The establishment of infection with Candida species appears to be a property of the host - not the organism. The more debilitated the host, the more invasive the disease. The clinical material to be sent to the lab depends on the presentation of the disease: Such material may include blood cultures, vaginal discharge, urine, feces, nail clippings or material from cutaneous or mucocutaneous lesions. Candida is a polymorphic yeast, i.e., yeast cells, hyphae and pseudohyphae are produced. It has been shown that Candida needs a transcription repressor to maintain the yeast form. This ability to assume various forms may be related to the pathogenicity of this organism. The yeast form is 10 to 12 microns in diameter, gram positive, and it grows overnight on most bacterial and fungal media. It also produces germ tubes (figure 9 and 10), and pseudohyphae (figure 6 and 7) may be formed from budding yeast cells that remain attached to each other. Spores may be formed on the pseudomycelium. These are called chlamydospores and they can be used to identify different species of Candida. Some mycologists think that the pseudomycelial form represents a more invasive form of the organism. The species are identified by biochemical reactions. The organism occurs world-wide. The drugs of choice for systemic infection are itraconazole and fluconazole. If an artificial heart valve or in-dwelling catheter becomes infected, it must be replaced. Drug therapy alone will not suppress the organism if the foreign body remains in the host. This resistance is due to biofilms which we will discuss later.
|
||||
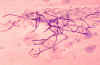
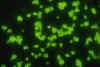 Figure
3 Figure
3Oval budding yeast cells of Candida albicans. Fluorescent antibody stain. CDC/Maxine Jalbert, Dr. Leo Kaufman. lek1@cdc.gov |
|
||||
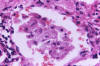
 Figure 8
Figure 8Histopathology of Candida esophagitis. Methenamine silver stain (digitally colorized). CDC |
|
||||
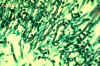
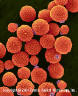 Figure 12
Figure 12Encapsulated pathogenic yeast fungus (Cryptococcus neoformans). A yeast-like fungus that reprouces by budding. A acidic mucopolysaccharide capsule completely encloses the fungus. It can cause the disease called cryptococcosis; especially in immune deficient humans, such as in patients with HIV / AIDS. The infection may cause meningitis in the lungs, skin or other body regions. The most common clinical form is meningoencephalitis. It is caused by inhaling the fungus found in soil that has been contaminated by pigeon droppings. © Dennis Kunkel Microscopy, Inc. Used with permission
|
CRYPTOCOCCOSIS
|
||||
|
|||||
|
|
|||||