|
x |
x |
|
 |
 |
|
INFECTIOUS
DISEASE |
BACTERIOLOGY |
IMMUNOLOGY |
MYCOLOGY |
PARASITOLOGY |
VIROLOGY |
|
VIETNAMESE |
IMMUNOLOGY - CHAPTER ELEVEN
RESPONSE TO ANTIGEN: PROCESSING AND PRESENTATION
MHC RESTRICTION AND ROLE OF THE THYMUS
Gene Mayer, Ph.D
Emertius Professor of Pathology, Microbiology and Immunology
University of South Carolina
|
|
TURKISH |
|
FRANCAIS |
|
PORTUGUES |
Let us know what you think
FEEDBACK |
|
SEARCH |
|
|

 |
|
Logo image © Jeffrey
Nelson, Rush University, Chicago, Illinois and
The MicrobeLibrary |
|
|
|
|
|
TEACHING
OBJECTIVES
To compare and contrast antigens recognized by the TCR and BCR.
To describe the pathways involved in processing endogenous and exogenous
antigens.
To discuss self MHC restriction in antigen presentation to T cells
To describe the major antigen presenting cells.
To compare and contrast presentation of conventional and superantigens.
To discuss the role of positive and negative selection in the thymus in
generation of self MHC restricted T cells.
KEY WORDS
Endogenous antigen
Class I antigen processing pathway
Proteosome
Transporter
Exogenous antigen
Class II antigen processing pathway
Invariant chain
Self MHC restriction
Positive selection
Negative selection
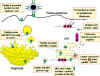 Figure 1
Figure 1
Pathway of class I MHC restricted presentation of an endogenously
synthesized antigen. An example of such an antigen would be a viral
protein made in the cell as a result of infection |
Comparison of BCR and TCR
B cells and T cells recognize
different substances as antigens and in a different form. The B
cell uses cell surface-bound immunoglobulin as a receptor and
the specificity of that receptor is the same as the
immunoglobulin that it is able to secrete after activation. B
cells recognize the following antigens in soluble form:
In contrast, the overwhelming
majority of antigens for T cells are proteins, and these must be
fragmented and recognized in association with MHC products
expressed on the surface of nucleated cells, not in a soluble
form. T cells are grouped functionally according to the class of MHC molecules that associate with the peptide fragments of
the protein: helper T cells recognize only those peptides associated
with class II MHC molecules, and cytotoxic T cells recognize
only those peptides associated with class I MHC molecules.
ANTIGEN PROCESSING AND PRESENTATION
Antigen processing and presentation are processes that occur within a cell
that result in fragmentation (proteolysis) of proteins, association of the
fragments with MHC molecules, and expression of the peptide-MHC molecules at
the cell surface where they can be recognized by the T cell receptor on a T
cell. However, the path leading to the association of protein fragments with
MHC molecules differs for class I and class II MHC. MHC class I molecules
present degradation products derived from intracellular (endogenous)
proteins in the cytosol. MHC class II molecules present fragments derived
from extracellular (exogenous) proteins that are located in an intracellular
compartment.
Antigen processing and presentation in cells expressing class I MHC
All nucleated cells express class I MHC. As shown in Figure 1, proteins are
fragmented in the cytosol by proteosomes (a complex of proteins having
proteolytic activity) or by other proteases. The fragments are then
transported across the membrane of the endoplasmic reticulum by transporter
proteins. (The transporter proteins and some components of the proteosome
have their genes in the MHC complex). Synthesis and assembly of class I
heavy chain and beta2 microglobulin occurs in the endoplasmic
reticulum. Within the endoplasmic reticulum, the MHC class I heavy chain,
beta2microglobulin and peptide form a stable complex that is
transported to the cell surface.
|
|
|
Antigen processing and presentation in cells
expressing class II MHC
Whereas all nucleated cells express class I MHC, only
a limited group of cells express class II MHC, which includes the antigen
presenting cells (APC). The principal APC are macrophages, dendritic
cells (Langerhans cells), and B cells, and the expression of
class II MHC molecules is either constitutive or inducible,
especially by interferon-gamma in the case of macrophages.
As shown in Figure 2, exogenous proteins taken in by
endocytosis are fragmented by proteases in an
endosome. The alpha and beta chains of MHC class II, along with an invariant chain, are
synthesized, assembled in the endoplasmic reticulum, and transported through
the Golgi and trans-Golgi apparatus to reach the endosome, where the
invariant chain is digested, and the peptide fragments from the exogenous
protein are able to associate with the class II MHC molecules, which are
finally transported to the cell surface.
|
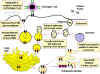 Figure 2 Figure 2
Pathway of class II MHC-restricted presentation of an exogenous
antigen |
|
|
Important aspects of antigen processing and
presentation
Viruses replicate within nucleated cells in the
cytosol and produce endogenous antigens that can associate with
class I MHC. By killing these infected cells, cytolytic T cells
help to control the spread of the virus.
Bacteria mainly reside and replicate extracellularly. By being taken up and fragmented inside cells as exogenous
antigens that can associate with class II MHC molecules, helper Th2 T
cells can be activated to assist B cells to make antibody against
bacteria, which limits the growth of these organisms.
Some bacteria grow intracellularly inside the vesicles
of cells like macrophages. Inflammatory Th1 T cells help to
activate macrophages to kill the intracellular bacteria.
-
Fragments of self, as well as non-self,
proteins associate with MHC molecules of both classes and are expressed at
the cell surface.
-
Which protein fragments bind is a function of the
chemical nature of the groove for that specific MHC molecule.
|
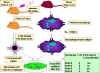 Figure 3
Figure 3
Self MHC Restriction of Th/APC Interactions |
 Figure 4
Figure 4
Virus-specific CTLs from a strain A or strain B mouse lyse only
syngeneic target cells infected with a specific virus. The CTLs do
not lyse uninfected target cells and are not alloreactive. Further
analysis has shown that the CTLs and target cells must come from
animals that share class I MHC alleles in order for the target to
present viral antigens to the CTLs. |
SELF MHC RESTRICTION
In order for a T cell to recognize and respond to a
foreign protein antigen, it must recognize the MHC on the presenting cell as
self MHC. This is termed self MHC restriction. Helper T cells
recognize antigen in context of class II self MHC. Cytolytic T cells
recognize antigen in context of class I self MHC. The process whereby
T cells become restricted to recognizing self MHC molecules occurs in the thymus.
The experimental systems demonstrating self MHC
restriction for APC-helper T cell interactions and for class I MHC-cytotoxic
T cell interactions are shown in Figures 3 and 4, respectively.
|
 Figure 5
Figure 5
Differences between antigen and super antigen. Antigenic peptides are
processed within the cell and presented on the cell surface in
association with class II MHC molecules. They then trigger the T-cell receptor
on a T lymphocyte. Superantigens are not processed but bind to the class
II MHC protein and to the V beta chain of the T cell receptor. A given
superantigen activates a distinct class of T cells that express a
certain V beta chain.
Note: In the case of MHC
II-TCR interaction
with a normally processed peptide, recognition of the peptide on the MHC
molecule requires V alpha, J alpha, V beta, D beta and J beta segments
of the TCR. Such an interaction occurs at low frequency. In the case of
MHC II-TCR interaction with an unprocessed superantigen, only a given V
beta region is recognized. This clearly would occur at a much higher
frequency |
Antigen Presenting Cells
The three main types of antigen presenting cells are dendritic cells,
macrophages and B cells, although other cells, that express class II MHC
molecules, (e.g., thymic epithelial cells) can act as antigen
presenting cells in some cases. Dendritic cells, which are found in skin
and other tissues, ingest antigens by
pinocytosis and transport antigens to
the lymph nodes and spleen. In the lymph nodes and spleen they are found
predominantly in the T cells areas. Dendritic cells are the most effective
antigen presenting cells and can present antigens to naïve (virgin) T
cells. Furthermore, they can present internalized antigens in association
with either class I or class II MHC molecules (cross presentation), although
the predominant pathway for internalized antigen is the class II pathway.
The second type of antigen presenting cell is the macrophage. These cells
ingest antigen by phagocytosis or pinocytosis. Macrophages are not as
effective in presenting antigen to naïve T cells but they are very good in
activating memory T cells. The third type of antigen presenting cell is the
B cell. These cells bind antigen via their surface immunoglobulin and ingest antigens
by pinocytosis. Like macrophages these cells are not as effective as
dendritic cells in presenting antigen to naïve T cells. B cells are very
effective in presenting antigen to memory T cells, especially when the
antigen concentration is low because surface immunoglobulin on the B cells binds antigen
with a high affinity.
Presentation of Superantigens
Superantigens are antigens that can polyclonally activate T cells (see
antigens) to produce large quantities of cytokines that can have
pathological effects. These antigens must be presented to T cells in
association with class II MHC molecules but the antigen does not need to be
processed. Figure 5 compares how conventional antigens and superantigens
are presented to T cells. In the case of a superantigen, the intact protein
binds to class II MHC molecules and to one or more Vβ regions of
the TCR. The antigen is not bound to the peptide binding groove of the MHC
molecule or to the antigen binding site of the TCR. Thus, any T cell that
uses a particular Vβ in its TCR will be activated by a
superantigen, resulting in the activation of a large numbers of T cells.
Each superantigen will bind to a different set of Vβ regions.
|
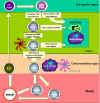 Figure 6
Figure 6
Prethymic T cells enter the thymus rudiment and proliferate as large
lymphoblasts in the sub-capsular region of the thymus. The
lymphoblasts replicate resulting in a pool of cells that
differentiate. Here the cells become CD8 and CD4 positive but expression is
low. TCR genes are also rearranged in these cells and the products may
also be expressed on the cell surface at low levels. As the cells
mature, they move into the cortex where they adhere to cortical
epithelial cells which are long and branched, providing a large
surface area to interact with other cells. TCRs on the surfaces of
thymocytes interact with the MHC molecules on the epithelial cells
leading to positive selection. The cells that are not selected are
subject to apoptosis and are phagocytosed by macrophages. As the
thymocytes migrate further into the cortex of the thymus, the
expression of CD3, CD4, CD8 and TCR increases. TCRs with
self-reactivity are deleted because of contact with autoantigens
presented by dendritic cells and macrophages. This leads to negative
selection. Cells that express CD4 or CD8 appear and
migrate to the periphery by specialized vessels in the cortico-medullar
region. |
THYMic education
Both Th and Tc cells are self-MHC
restricted. In addition, T cells do not normally recognize self antigens.
How are self MHC restricted T cells generated and why are self reacting T
cells not produced? Random VDJ rearrangements in T cells would be expected
to generate some T cells that can recognize non-self MHC and some T cells
that can recognize self antigens. It is the role of the thymus to ensure
that the only T cells that get to the periphery are self-MHC restricted and
unable to react with self antigen. Functional T cells in the periphery have
to recognize foreign antigens associated with self MHC, because APC or
target cells present foreign antigen associated with self MHC. However, an
individual does not need functional T cells in the periphery that recognize
antigen (self or foreign) associated with foreign MHC. An individual
especially does not want functional T cells in the periphery that can
recognize self antigens associated with self MHC because they could lead to
damage of healthy, normal tissues.
As a result of random VDJ recombination events occurring in immature T cells
within the thymus, TCRs of all specificities are produced. Processes in the
thymus determine which TCR specificities are retained. There are two
sequential steps shown in Figure 6. First, T cells with the ability to bind
to self MHC molecules expressed by cortical thymic epithelial cells are
retained. This is known as positive selection. Those that do not bind,
undergo apoptosis. Thus, T cells having a TCR that recognizes self MHC
survive. Next, T cells with the ability to bind to self MHC molecules
associated with self molecules expressed by thymic epithelial cells,
dendritic cells and macrophages are killed. This is known as negative
selection. Those that do not bind are retained. As a result of these two
steps, T cells having a TCR that recognizes self MHC and foreign antigen
survive. Each T cell that survives positive and negative selection in the
thymus and is released into the periphery retains its specific T cell
receptor.
While positive and negative selection is occurring in the thymus the
immature T cells are also expressing CD4 or CD8 antigens on their surface.
Initially the pre-T cell that enters the thymus is CD4-CD8-. In the thymus
it becomes CD4+CD8+ and as positive and negative selection proceeds a cell
becomes either a CD4+ or CD8+ cell. The commitment to become either a CD4+
or CD8+ cells depends on which class of MHC molecule the cell encounters. If
a CD4+CD8+ cell is presented with a class I molecule it will down regulate
CD4 and become a CD8+ cell. If a cell is presented with a class II MHC
molecule it will down regulate CD8 and become a CD4+ cell (Figure 7).
Negative selection in the
periphery
Positive and negative
selection in the thymus is not a 100% efficient process. In addition,
not all self antigens may be expressed in the thymus. Thus some self
reactive T cells may get to the periphery. Thus, there are additional
mechanisms that are designed to eliminate self reactive T cells in the
periphery. These will be discussed in the
tolerance
chapter.
B CELL SELECTION
Since B cells are not MHC-restricted there is no need for positive
selection of B cells. However, negative selection (i.e., elimination of
self-reactive clones) of B cells is required. This occurs during B cell
development in the bone marrow. However, negative selection of B cells
is not a critical as for T cells since, in most instances, B cells
require T cell help in order to become activated. Thus, if a self
reactive B cell does get to the periphery it will not be activated due
to lack of T cell help.
|
| |
 Figure 7
Figure 7
CD4- CD8- precursor thymocytes become double positive, CD4+ CD8+ cells expressing low levels of the alpha and beta
chains of the T cell receptor (TCR). Positive selection for
interaction with self MHC-I or MHC-II molecules occurs in the cortical
epithelium. The majority of the cells are unselected and undergo
apoptosis. The cells that remain either interact with MHC-I and
lose their CD4 antigen or interact with MHC-II and lose their CD8
antigen. Autoreactive cells are then removed as a result of their
interaction with self antigen peptides that are presented by cells in
the corticomedullary junction and the medulla of the thymus
|
| |
|
| |
 Return to the Immunology Section of Microbiology and Immunology On-line
Return to the Immunology Section of Microbiology and Immunology On-line
|
|
|
This page last changed on
Monday, September 18, 2017
Page maintained by Richard Hunt
|

 Figure 2
Figure 2 Figure 3
Figure 3 Figure 4
Figure 4 Figure 5
Figure 5 Figure 6
Figure 6
