|
x |
x |
|
 |
 |
|
INFECTIOUS
DISEASE |
BACTERIOLOGY |
IMMUNOLOGY |
MYCOLOGY |
PARASITOLOGY |
VIROLOGY |
|
VIETNAMESE
|
IMMUNOLOGY - CHAPTER TWELVE
CELL-MEDIATED IMMUNITY:
Cell-cell interactions in specific immune responses
Dr Gene Mayer
Professor
Department of Pathology, Microbiology and Immunology
University of South Carolina School of Medicine
and
Dr Jennifer Nyland
Assistant Professor
Department of Pathology, Microbiology and Immunology
University of South Carolina School of Medicine
|
|
TURKISH |
|
FRANCAIS |
|
PORTUGUES |
|
Let us know what you think
FEEDBACK |
|
SEARCH |
|
|
|

 |
|
Logo image © Jeffrey
Nelson, Rush University, Chicago, Illinois and
The MicrobeLibrary |
|
|
|
Edited and
illustrated by Dr Richard Hunt
|
|
|
|
TEACHING
OBJECTIVES
To discuss the central role of Th cells in immune responses
To describe the cell-cell interactions which occur in (i) antibody
responses to T-dependent antigens, (ii) generation of cytotoxic T cells,
and (iii) activation of macrophages and NK cells
To discuss the mechanisms of killing by cytotoxic T cells and NK
cells
To discuss responses to T-independent antigens.
|
Central role of Th cells in immune responses
As
depicted in Figure 1, after Th cells recognize specific
antigen presented by an
antigen-presenting cell (APC), they can initiate several key immune processes. These include:
-
Selection of appropriate effector mechanisms ( e.g., B cell activation or
Tc generation);
-
Induction of proliferation of appropriate effector
cells
-
Enhancement of the functional activities of other cells (e.g.,
granulocytes, macrophages, NK cells).
There
are four subpopulations of Th cells: Th0, Th1, Th2 and Th17 cells. When naïve Th0
cells encounter antigen in secondary lymphoid tissues, they are capable of
differentiating into inflammatory Th1 cells, helper Th2 cells or pathogenic T17
cells, which are
distinguished by the cytokines they produce (Figure 2). Whether a Th0 cells
becomes a Th1, a Th2 or a T17 cell depends upon the cytokines in the environment, which
is influenced by antigen. For example some antigens stimulate IL-4 production
which favors the generation of Th2 cells while other antigens stimulate IL-12
production, which favors the generation of Th1 cells. Th1, Th2 and Th17 cells affect
different cells and influence the type of an immune response, as shown in Figure
3 for Th1 and Th2 cells.
Cytokines produced by Th1 cells activate macrophages and participate in the
generation of cytoxic lymphocytes (CTL), resulting in a cell-mediated immune response. In
contrast cytokines produced by Th2 cells help to activate B cells, resulting in
antibody production. In a relatively recent discovery, Th17 cells (designated as such by their
production of IL-17) differentiate (in humans) in response to IL-1, IL-6, and
IL-23. TGF-β is important for Th17 differentiation in mice, but not in humans.
IL-17 enhances the severity of some autoimmune diseases including multiple
sclerosis, inflammatory bowel disease, and rheumatoid arthritis.
Equally important, each subpopulation can exert inhibitory influences on the
other. IFN-γ produced by Th1 cells inhibits proliferation of Th2 cells
and differentiation of Th17 cells and
IL-10 produced by Th2 cells inhibits production of IFN-γ by Th1 cells. In
addition, although not shown, IL-4 inhibits production of Th1 cells and
differentiation of Th17 cells. Thus, the
immune response is directed to the type of response that is required to deal
with the pathogen encountered – cell-mediated responses for intracellular
pathogens or antibody responses for extracellular pathogens.
|
|
KEY WORDS
Th1 cells
Th2 cells
Hapten-carrier model
CD28
B7
CD40
CD40 ligand
CD5
B1 cells
B2 cells
CTL
Fas ligand
Perforin
Granzymes
Caspases
IFN-γ
Activation |
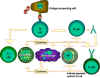 Figure 1
Figure 1
Th cells are at the center of cell-mediated immunity. The
antigen-presenting cells present antigen to the T helper (Th) cell.
The Th cell recognises specific epitopes which are selected as
target epitopes. Appropriate effector mechanisms are now determined.
For example, Th cells help the B cells to make antibody and also
activate other cells. The activation signals produced by Th cells
are cytokines (lymphokines) but similar cytokines made by
macrophages and other cells also participate in this process
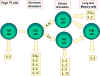 Figure 2
Figure 2
Differentiation of murine Th cells.
Mouse Th cells differentiate into subsets that synthesize different
patterns of lymphokines. This also occurs in humans
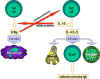 Figure 3
Figure 3
Selection of effector mechanisms by Th1 and Th2 cells.
In addition to determining various effector pathways by virtue of
their lymphokine production, Th1 cells switch off Th2 cells and
vice versa
|
|
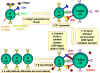 Figure 4
Figure 4
Molecules involved in the interactions of B and TH cells
Antigen  is processed by B cell.
Co-stimulators are expressed. The processed antigen peptide is processed by B cell.
Co-stimulators are expressed. The processed antigen peptide
 is presented in association with MHC class II antigens. The T cell
recognizes the peptide along with the MHC antigen and the
co-stimulators. The T cell expresses CD40 ligand. The latter binds
to CD40 antigen on the B cell and the B cells divide and
differentiate. Antibodies are produced by the B cell
is presented in association with MHC class II antigens. The T cell
recognizes the peptide along with the MHC antigen and the
co-stimulators. The T cell expresses CD40 ligand. The latter binds
to CD40 antigen on the B cell and the B cells divide and
differentiate. Antibodies are produced by the B cell
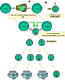 Figure 5
Figure 5
Cooperation of cells in the immune response
Antigen-presenting cells (e.g. dendritic cells) present processed
antigen to virgin T cells, thereby priming them. B cells also process
the antigen and present it to the T cells. They then receive signals
from the T cells that cause them to divide and differentiate. Some B
cells form antibody-forming cells while a few form B memory cells |
Cell-cell interactions in antibody responses to
exogenous T-dependent antigens
Hapten-carrier model
Historically one of the major findings in
immunology was that both T cells and B cells were required for antibody
production to a complex protein. A major contribution to our
understanding of this process came from studies on the formation of
anti-hapten antibodies. Studies with hapten-carrier conjugates
established that:
-
Th2 cells recognized the carrier determinants and B
cells recognized haptenic determinants
-
Interactions between hapten-specific
B cells and carrier-specific Th cells was self MHC restricted
-
B cells can function both in antigen recognition and in
antigen presentation
B cells occupy a unique position in immune responses
because they express immunoglobulin and class II MHC molecules on their cell
surface. They therefore are capable of producing antibody having the
same specificity as that expressed by their immunoglobulin receptor; in
addition they can function as an antigen presenting cell. In terms of
the hapten-carrier conjugate model, the mechanism is thought to be the
following: The hapten is recognized by the immunoglobulin receptor, the hapten-carrier
is brought into the B cell, processed, and peptide fragments of the
carrier protein are presented to a helper T cell. Activation of the T
cell results in the production of cytokines that enable the hapten-specific
B cell to become activated to produce soluble anti-hapten antibodies.
Figure 4 summarizes the B cell-T cell interactions that occur.
Note that there are multiple signals delivered to the B cells in this
model of Th2 cell-B cell interaction. As was the case for activation of
T cells where the signal derived from the TCR recognition of a peptide-MHC
molecule was by itself insufficient for T cell activation, so too for
the B cell. Binding of an antigen to the immunoglobulin receptor
delivers one signal to the B cell, but that is insufficient. Second
signals delivered by co-stimulatory molecules are required; the most
important of these is CD40L on the T cell that binds to CD40 on the B
cell to initiate delivery of a second signal.
Cell-cell
interactions in the primary antibody response
B cells are not the best antigen
presenting cell in a primary antibody response; dendritic cells or
macrophages are more efficient. Nevertheless, with some minor
modifications the hapten-carrier model of cell-cell interactions
described above also applies to interactions in a primary antibody
response (Figure 5). In a primary response the Th2 cell first encounters
antigen presented by dendritic cells or macrophages. The “primed” Th2
cell can then interact with B cells that have encountered antigen and
are presenting antigenic peptides in association with class II MHC
molecules. The B cells still require two signals for activation – one
signal is the binding of antigen to the surface immunoglobulin and the second signal
comes from CD40/CD40 ligand engagement during Th2/B cell-cell
interaction. In addition, cytokines produced by the Th2 cells help B
cells proliferate and differentiate into antibody secreting plasma
cells.
Cell-cell
interactions in the secondary antibody responses
As a consequence of a primary
response, many memory T and B cells are produced. Memory B cells have a
high affinity immunoglobulin receptor (due to affinity maturation), which allows
them to bind and present antigen at much lower concentrations than that
required for macrophages or dendritic cells. In addition, memory T cells
are more easily activated than naïve T cells. Thus, B/Th cell
interactions are sufficient to generate secondary antibody responses. It
is not necessary (although it can occur) to “prime” memory Th cells with
antigen presented by dendritic cells or macrophages.
Cytokines and class switching
Cytokines produced by activated Th2 cells not only
stimulate proliferation and differentiation of B cells, they also help
regulate the class of antibody produced. Different cytokines influence
the switch to different classes of antibodies with different effort
functions. In this way the antibody response is tailored to suit the
pathogen encountered (e.g. IgE antibodies for parasitic worm
infections). Table 1 shows the effects of different cytokines on the
class of antibody produced.
|
Cytokine
|
IgG1
|
IgG2a
|
IgG2b
|
IgG3
|
IgA
|
IgE
|
IgM
|
|
IL-4
|
Induce
|
Inhibit
|
|
Inhibit
|
|
Induce
|
Inhibit
|
|
IL-5
|
|
|
|
|
Augment
production
|
|
|
|
IFN-gamma
|
Inhibit
|
Induce
|
|
Induce
|
|
Inhibit
|
Inhibit
|
|
TGF-beta
|
|
|
Induce
|
Inhibit
|
Induce
|
|
Inhibit
|
|
Isotype
regulation by murine T cell cytokines.
Certain cytokines either induce (green) or inhibit (pink) the production
of certain antibody isotypes. Inhibition mostly results from switch to
the different isotype
|
|
Table 1 |
|
| |
Cell-cell interactions in antibody responses to exogenous T-independent
antigens
Antibody responses to
T-independent antigens do not require cell-cell interactions. The
polymeric nature of these antigens allows cross-linking
of antigen receptors on B cells resulting in activation. No secondary
responses, affinity maturation or class switching occurs. Responses to
T-independent antigens are due to the activation of a subpopulation of B cells
called CD5+ B cells (also called B1 cells), which distinguishes them from
conventional B cells that are CD5- (also called B2 cells).
CD5+ (B1) cells
CD5+ cells are the first B cells to appear in
ontogeny. They express surface IgM but little or no IgD and they produce
primarily IgM antibodies from minimally somatically mutated germ line genes.
Antibodies produced by these cells are of low affinity and are often
polyreactive (bind multiple antigens). Most of the IgM in serum is derived from
CD5+ B cells. CD5+ B cells do not give rise to memory cells. An important
characteristic of these cells is that they are self-renewing, unlike
conventional B cells which must be replaced from the bone marrow. CD5+ B cells
are found in peripheral tissues and are the predominant B cell in the peritoneal
cavity. B1 cells are a major defense against many bacterial pathogens that
characteristically have polysaccharides in their cell walls. The importance of
these cells in immunity is illustrated by the fact that many individuals with T
cell defects are still able to resist many bacterial pathogens.
|
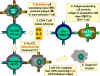 Figure 6
Figure 6
CTL cells must differentiate in response to antigen. In order to
differentiate into functional cytotoxic T lymphocytes, pre-CD8+ CTLs must
receive two different signals. First, they must recognize antigen presented
by MHC-I expressing cells (the stimulator cells) and, second, they must be
stimulated by cytokines. IL-2, interferon-gamma and others are
made by CD4+ helper T cells as a result of their interaction with class II
MHC-expressing antigen presenting cells. As a result of these two signals,
the pre-CTL differentiates into an active CTL that can then lyse target
cells that bear the same antigen.
Adapted
from Abbas, et. al. Cellular
and Molecular Immunology. 3rd Ed., p. 292.
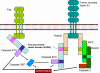 Figure 7
Figure 7
Fas- and TNF-mediated killing of target cells by CTLs
|
Cell-cell interactions in cell-mediated immunity - generation of Tc cells in
response to endogenous antigens in the cytosol
Cytotoxic T lymphocytes are not fully mature when they exit the thymus. They
have a functional TCR that recognizes antigen, but they cannot lyse a target
cell. They must differentiate into fully functional effector Tc cells.
Cytotoxic cells differentiate from a "pre-CTL" in response to two signals:
-
Specific antigen
in the context of class I MHC,
on a stimulator cell
-
Cytokines produced by Th1 cells, especially IL-2, and IFN-gamma. This is
shown in Figure 6.
Features
of CTL-mediated lysis
-
CTL killing is antigen-specific. To be killed by a CTL, the target
cell must bear the same class I MHC-associated antigen that triggered pre-CTL
differentiation.
-
CTL killing requires cell contact. CTL are triggered to kill when
they recognize the target antigen associated with a cell surface MHC molecule.
Adjacent cells lacking the appropriate target MHC-antigen are not affected.
-
CTLs are not injured when they lyse target cells. Each CTL is capable
of killing sequentially numerous target cells.
Mechanisms
of CTL-mediated killing
CTLs utilize several mechanisms to kill target cells, some of which require
direct cell-cell contact and others that result from the production of certain
cytokines. In all cases death of the target cells is a result of
apoptosis.
-
Fas- and TNF-mediated
killing (Figure 7)
Once generated CTLs express Fas
ligand on their surface, which binds to Fas receptors on target cells. In
addition, TNF-α secreted by CTLs can bind to TNF receptors on target cells. The
Fas and TNF receptors are a closely related family of receptors, which when they
encounter their ligands, for trimers of the receptors. These receptors also
contain death domains in the cytoplasmic portion of the receptor, which after
trimerization can activate caspases that induce apoptosis in the target cell.
-
Granule-mediated killing (Figure 8)
Fully differentiated CTLs have
numerous granules that contain
perforin and
granzymes. Upon contact with target
cells, perforin is released and it polymerizes to form channels in the target
cell membrane. Granzymes, which are serine proteases, enter the target cell
through the channels and activate caspases and nucleases in the target cell
resulting in apoptosis.
|
| |
Figure 8
Mechanisms for the CTL destruction of target cells
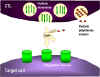 1. CTL degranulates and releases perforin monomers into the surroundings.
Enzymes that polymerize perforin to form polyperforin channels are also
released and these along with Ca++ catalyze channel formation in the
membrane of the target cell
1. CTL degranulates and releases perforin monomers into the surroundings.
Enzymes that polymerize perforin to form polyperforin channels are also
released and these along with Ca++ catalyze channel formation in the
membrane of the target cell
 2. The CTL may also release degradative enzymes and toxins which travel
through the perforin chanels and damage the target cell
2. The CTL may also release degradative enzymes and toxins which travel
through the perforin chanels and damage the target cell
 3. Cytokines such as TNF alpha and TNF beta are released from the CTL or
nearby macrophages. Interferon gamma may also be released from the CTLs or
from other nearby lymphoid cells. These bind to receptors on the target
cell and trigger apoptosis
3. Cytokines such as TNF alpha and TNF beta are released from the CTL or
nearby macrophages. Interferon gamma may also be released from the CTLs or
from other nearby lymphoid cells. These bind to receptors on the target
cell and trigger apoptosis |
|
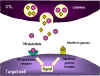
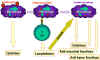 Figure 9
Figure 9
Macrophages play a central role in the immune system. before T and
B-cell immunity starts. Macrophages process antigens and present
them to T cells which then release lymphokines which activate the
macrophages to perform various other functions including the
production of more cytokines
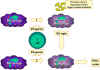 Figure 10
Figure 10
Macrophage activation results from the interaction of multiple
cytokines and other factors.
In pathway 1, TNF-alpha is released from macrophages as a result of
activation by interferon gamma and interaction with bacterial
components that trigger cytokine production. An example of such a
triggering component is bacterial lipopolysaccharide. The TNF-alpha
from pathway 1 leads to the production of nitric oxide by the
interferon-activated macrophage in pathway 2.
|
Cell-cell interactions in cell-mediated immunity
- activation of
macrophages in response to endogenous antigens in vesicles
Macrophages play a central role in the immune system. As shown in
Figure 9, macrophages are involved in:
-
Initial defense as part of the innate
immune system
-
Antigen presentation to Th cells
-
Various effector functions (e.g., cytokine production,
bactericidal and tumoricidal activities).
Indeed macrophages play an
important role not only in immunity but also in reorganization of
tissues. However, because of their potent activities, macrophage can
also do damage to tissues. Table 2 summarizes the many functions of
macrophages in immunity and inflammation.
Production
of:
IL-6, TNF alpha, IL-1 – act as pyrogen
|
Hydrolases
Hydrogen peroxide production
Complement C3a
TNF alpha production
|
Selection
of lymphocytes to be activated:
IL-12 results in Th1 activation
IL-10 results in Th2 activation
Activation
of lymphocytes:
Production of IL-1
Processing and presentation of antigen
|
Oxygen
–dependent production of:
hydrogen peroxide
superoxide
hydroxyl
radical
hypochlorous
acid
Oxygen-independent
production of:
acid hydrolases
cationic proteins
lysozyme
|
Reorganization of
tissues
Secretion
of a variety of factors:
Degradative enzymes (elastase,
hyaluronidase,collagenase)
Fibroblast stimulation factors
Stimulation of angiogenesis
|
Toxic
factors
Hydrogen peroxide
Complement C3a
Proteases
Arginase
Nitric oxide
TNF alpha
|
|
Table 2 |
Many of these macrophage functions can only be performed by
activated macrophages. Macrophage activation can be
defined as quantitative alterations in the expression of various gene
products that enable the activated macrophage to perform some function
that cannot be performed by the resting macrophage.
Macrophage activation is an important function of Th1 cells.
When Th1 cells get activated by an APC such as a macrophage, they
releases IFN-γ, which is one of two signals required to activate
a macrophage. Lipopolysaccharide (LPS) from bacteria or TNF-α
produced by macrophages exposed to bacterial products deliver the second
signal (Figure 10).
Effector mechanisms employed by macrophages include production of:
-
TNF-α, which can induce apoptosis
-
Nitric oxide and other reactive
nitrogen intermediates
-
Reactive oxygen intermediates
-
Cationic proteins and hydrolytic
enzymes
Macrophage activation by Th1 cells is very important in protection
against many different pathogens For example, Pneumocystis carinii,
an extracellular pathogen, is controlled in normal individuals by
activated macrophages; it is, however, a common cause of death in AIDS
patients because they are deficient in Th1 cells. Similarly, Mycobacterium tuberculosis,
an intracellular pathogen that resides in vesicles, is not efficiently
killed by macrophages unless they are activated; hence this infection is
a problem in AIDS patients.
|
| |
Cell-cell interactions in cell-mediated immunity
- activation of NK
cells
Cytokines produced by activated Th1 cells, particularly Il-2 and IFN-γ, also activate NK cells to become lymphokine activated killer
cells (LAK cells). LAK cells are able to kill virus infected and tumor
cells in a non-MHC-restricted manner. Indeed, susceptibility of target
cells to killing by NK and LAK cells is inversely proportional to the
expression of MHC class I molecules (see lecture on innate immunity).
The effector mechanisms used by NK and LAK cells to kill target cells is
similar to those used by CTLs (e.g., perforin and granzymes). NK
and LAK cells are also able to kill antibody coated target cells by
ADCC.
|
|
|
|
|

 |
 Return to the Immunology Section of Microbiology and Immunology On-line Return to the Immunology Section of Microbiology and Immunology On-line
This page last changed on
Monday, September 18, 2017
Page maintained by
Richard Hunt
|

 Figure 6
Figure 6










