|
x |
x |
|
 |
 |
|
INFECTIOUS
DISEASE |
BACTERIOLOGY |
IMMUNOLOGY |
MYCOLOGY |
PARASITOLOGY |
VIROLOGY |
|
SHQIP - ALBANIAN |
BACTERIOLOGY - CHAPTER SEVENTEEN
ZOONOSES
LISTERIA, FRANCISELLA,
BRUCELLA, BACILLUS, YERSINIA AND ERYSIPELOTHRIX
Dr Abdul Ghaffar
Professor Emeritus
University of South Carolina School of Medicine
|
|
EN ESPANOL - SPANISH |
|
Let us know what you think
FEEDBACK |
|
SEARCH |
|
|
|
|
|
|
|

 |
|
Logo image © Jeffrey
Nelson, Rush University, Chicago, Illinois and
The MicrobeLibrary |
|
|
|
 Arnold Boecklin:
The Plague 1898. Tempera on wood, Kunstmuseum, Basel
Arnold Boecklin:
The Plague 1898. Tempera on wood, Kunstmuseum, Basel |
|
TEACHING
OBJECTIVES
To know the general morphology
and physiology the organisms
To know epidemiology and clinical
symptoms
To understand the mechanisms
pathogenesis
To know the diagnostic, therapeutic
and preventive procedures
|
Zoonosis
refers to a disease primarily of animals which can be transmitted to humans as a
result of direct or indirect contact with infected animal populations.
BRUCELLOSIS
Morphology and physiology
Brucella are Gram-negative, non-motile, coccobacilli. They
are strict aerobes and grow very slowly (fastidious) on blood agar. In
the host, they live as facultative intracellular pathogens.
Epidemiology
Brucellosis is primarily a
disease of animals and it affects organs rich in the sugar
erythritol (breast,
uterus, epididymis, etc.). The organisms localize in these animal organs and
cause infertility, sterility,
mastitis, abortion.
They may also be carried asymptomatically. Humans
in close contact with infected animals (slaughterhouse workers, veterinarians,
farmers, dairy workers) are at risk of developing undulant fever. There
are 100 to 200 cases of brucellosis seen in the United States annually, although the worldwide
incidence is estimated at 500,000. Species of Brucella that are
known to infect humans include:
There are other species of Brucella that infect many
animals from whales to rats
Although brucellosis
has largely been eradicated in most developed countries through animal vaccination, it
persists in many underdeveloped and developing countries.
Ares where there is a higher risk of Brucella
infection include:
|
|
|
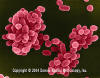 Brucella abortus - Gram-negative, coccobacillus prokaryote; causes bovine spontaneous abortion due to its rapid growth in the presence of
erythritol (produced in the plancenta). SEM x 29,650
©
Dennis Kunkel Microscopy, Inc.
Used with permission
Brucella abortus - Gram-negative, coccobacillus prokaryote; causes bovine spontaneous abortion due to its rapid growth in the presence of
erythritol (produced in the plancenta). SEM x 29,650
©
Dennis Kunkel Microscopy, Inc.
Used with permission
 Brucella spp. are poorly staining, small gram-negative coccobacilli (0.5-0.7 x 0.6-1.5
micrometer), and are seen mostly as single cells and appearing like “fine sand”
CDC
Brucella spp. are poorly staining, small gram-negative coccobacilli (0.5-0.7 x 0.6-1.5
micrometer), and are seen mostly as single cells and appearing like “fine sand”
CDC
 Brucella spp. Colony Characteristics: A. Fastidious, usually not visible at 24h. B. Grows slowly on most standard laboratory media (e.g. sheep blood, chocolate and trypticase soy agars). Pinpoint, smooth, entire translucent,
Brucella spp. Colony Characteristics: A. Fastidious, usually not visible at 24h. B. Grows slowly on most standard laboratory media (e.g. sheep blood, chocolate and trypticase soy agars). Pinpoint, smooth, entire translucent,
non-hemolytic at 48h.
CDC
 Brucella ovis in
epididymis © Bristol Biomedical Image Archive, University of
Bristol. Used with permission
Brucella ovis in
epididymis © Bristol Biomedical Image Archive, University of
Bristol. Used with permission |
Transmission
Infection
may occur by:
-
Direct contact with infected
animals or animal products
-
Ingestion of animal products
such as unpasteurized milk, milk products (including cheeses),
undercooked meat. This is probably the most frequent cause of human
Brucella infections and are of particularly important for tourists
-
Breathing in bacteria/ This may occur in people in
contact with infected animals or in laboratories where Brucella
is handled.
-
Entry through wounds or mucous membranes. In addition,
to those mentioned above, this may occur in hunters. Infected game
animals may include (CDC): Bison, elk, caribou, moose, feral hogs.
-
Breast feeding. Person to person transmission is very
rare but has been reported
-
Sexual activity. Again this is very rare
-
Possibly in tissue transplantation or organ transplant.
Again, this is very rare.
CDC notes that it is especially important that expectant
mothers who have been exposed to Brucella should consult a physician
as post-exposure prophylaxis may be required.
The bacteria are engulfed by neutrophils and
monocytes and localize in the regional lymph nodes, where they proliferate
intracellularly. If the Brucella organisms are not destroyed or contained in the
lymph nodes, the bacteria are released from the lymph nodes resulting in
septicemia. The organisms migrate to other lympho-reticular organs (spleen, bone
marrow, liver, testes) producing granulomas and/or micro abscesses.
Symptoms
B. abortus
and B. canis cause a mild suppurative febrile infection whereas B.
suis causes a more severe
suppurative infection which can lead to
destruction of the lymphoreticular organs and kidney. B. melitensis is
the cause of most severe prolonged recurring disease. The bacteria enter the
human host through the mucous membranes of the oropharynx (ingestion/inhalation
routes), through abraded skin, or through the conjunctiva.
Symptoms
include:
Brucellosis may be either acute or
chronic.
Mortality due to Brucellosis is
rare (less than 3%) and is generally due to endocarditis.
Pathogenesis
The symptoms of brucellosis are due to the presence of the organism and appear
2 to 4 weeks (sometimes up to 2 months) after exposure. While in the phago-lysosome,
B. abortus releases 5'-guanosine and adenine which are capable of
inhibiting the degranulation of peroxidase-containing granules and thus inhibit
the myeloperoxidase-peroxide-halide system of bacterial killing. The
intracellular persistence of bacteria results in granuloma formation in the
reticuloendothelial system organs and tissue damage due to hypersensitivity
reactions, mostly type-IV.
Diagnosis
Diagnosis is based on prolonged (at least a week) presence of undulating fever,
myalgia,
arthralgia and the history of exposure (contact with animals or
consumption of unprocessed material from infected animals). Definitive diagnosis
can be made by culturing blood samples on blood enriched media. The (fastidious)
organisms grow very slowly (4 to 6 weeks in blood culture). B. abortus but
not other Brucella grow better in 5% CO2 atmosphere. On blood
agar, they produce white glistening colonies. Serology can be used to further
confirm the diagnosis.
Prevention and treatment
Prolonged (6 to 8 weeks) treatment with rifampin
or doxycycline along with streptomycin or tetracyclin is used to treat human Brucella infections.
Control
measures include animal vaccination and avoidance of infected material.
|
|
|
|
 Dr. Alexandre Yersin in Front of the National Quarantine Station, Shanghai
Station, 1936
Dr. Alexandre Yersin in Front of the National Quarantine Station, Shanghai
Station, 1936
 World distribution of plague 1998
CDC
World distribution of plague 1998
CDC
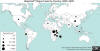
World distribution of plague 2000-2009
CDC
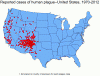
US distribution of plague by county
1970-2012
CDC
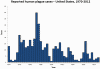
Cases of plague in the United States by year. 1970-2012
CDC
 Yersinia pestis - rod prokaryote (dividing); causes bubonic plague (SEM x20,800)
©
Dennis Kunkel Microscopy, Inc.
Used with permission
Yersinia pestis - rod prokaryote (dividing); causes bubonic plague (SEM x20,800)
©
Dennis Kunkel Microscopy, Inc.
Used with permission
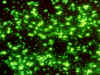 Yersinia pestis. Fluorescent antibody
identification CDC Yersinia pestis. Fluorescent antibody
identification CDC
|
PLAGUE
Plague is caused by Yersinia
pestis and is the disease known in the middle ages as the black death.
This is because it frequently leads to gangrene and blackening of various
parts of the body. Capillary fragility results in hemorrhages in the skin
which also result in black patches.
Morphology and physiology
Yersinia pestis is a
pleomorphic, Gram-negative, bipolar staining,
facultatively aerobic, non-motile bacillus. Optimal temperature for growth is 28
degrees C. It is a facultative intracellular parasite.
Epidemiology, transmission and
symptoms
The three documented pandemics of
plague (Black Death) have been responsible for the death of hundreds of millions
of people. Today, sporadic infections still occur. In the U.S., animal (sylvatic)
plague occurs in a number of western states, usually in small rodents and in
carnivores which feed on these rodents. The last urban outbreak of plague
occurred in the United States in 1924-25. There was an outbreak of
plague in India in 1994. In both cases, rats were the vector.
Sporadic plague still occurs in the rural United States.
Infected animals include:
-
squirrels (rock and ground)
-
wood rates
-
prairie dogs
-
mice
-
chipmunks
-
voles
-
rabbits
-
also carnivores may be infected by eating infected
prey. In the United States, pneumonic plague has occurred in recent
years from contact with infected cats that have been infected after
eating infected rodents.
Humans are usually infected by carrier
rodent fleas. The flea acquires the Y.
pestis organisms during a blood meal from infected rodents. The bacteria
lose their capsule, multiply in the intestinal tract and partially block the proventriculus.
When the flea feeds on a human host, it may regurgitate some of
the organisms into the wound.
Humans can also can be infected in other ways such as by
contact with infected animals or handling carcasses of infected animals.
This usually results in bubonic or septicemic plague.
Plague can also be spread in aerosols in a cough or
sneeze from a person (or pet cat) with pneumonic plague.
The bulk of non-capsular organisms are phagocytized and destroyed by neutrophils. However,
a few organisms are taken up
by histiocytes which are unable to kill them and allow them to resynthesize
their capsule and multiply. The encapsulated organisms, when they are released
from histiocytes, are resistant to phagocytosis and killing by neutrophils. The
resulting infection spreads to the draining lymph nodes which become hot,
swollen, tender and hemorrhagic giving rise to the characteristic black buboes
whence the name of the disease, bubonic plague, is derived. Within
hours the organism spreads into the spleen, liver and lungs resulting in
pneumonia. While in the circulation, the organism causes diffuse intra-vascular
coagulation resulting in intra-vascular thrombi and purpuric lesions all over the
body. If untreated, the infection has a very high (up to 90%) mortality rate. The
organisms in exhaled in cough droplets, infect other humans in close proximity
and cause pneumonic plague, which is more difficult to control and has 100%
mortality.
Pathogenesis
Many factors play direct and indirect roles in
Y. pestis
pathogenesis.
Low calcium response (lcr)
This is a plasmid-coded gene that
enables the organism to grow in a low Ca++ (intracellular) environment. It
also coordinates the production of several other virulence factors, such as
V, W and yops (Yersinia outer proteins).
V and W proteins
These plasmid-coded proteins are associated rapid proliferation and
septicemia.
Yops
A group of 11 proteins, which are coded by plasmids, are essential for
rodent pathogenesis and are responsible for cytotoxicity, inhibition of
phagocyte migration and engulfment and platelet aggregation.
Envelope (F-1) antigen
This is a protein-polysaccharide complex which is highly expressed at 37
degrees in
the mammalian host but not in the flea and is anti-phagocytic.
Coagulase and Plasminogen
activator
Both of these are plasmid-coded proteins.
Coagulase is responsible for micro thrombi formation
and
plasminogen activator
promotes the dissemination of the organism. It
also destroys C3b on the bacterial surface, thus attenuating phagocytosis.
|
|
|
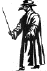
 Plague suits. The beak was filled with sweet smelling oils and vinegar
to counteract the smell of plague victims. Although physicians in the 1300's
did not know the cause of plague, the suits may have been effective to some
degree in that they kept fleas off and the shiny beak may have posed an
obstacle to the entry of fleas. Left From:
Bubonic Plague by Ely Janis Right:
©
Bristol Biomedical Image Archive, University of Bristol. Used with
permission
Plague suits. The beak was filled with sweet smelling oils and vinegar
to counteract the smell of plague victims. Although physicians in the 1300's
did not know the cause of plague, the suits may have been effective to some
degree in that they kept fleas off and the shiny beak may have posed an
obstacle to the entry of fleas. Left From:
Bubonic Plague by Ely Janis Right:
©
Bristol Biomedical Image Archive, University of Bristol. Used with
permission
|
Diagnosis
Diagnosis is based on appearance of buboes
(swollen lymph glands). The diagnosis is confirmed by
culture of a lymph node aspirate. Extreme caution is warranted in handling of
the specimen, as it is highly infectious. In cases of pneumonic and septicemic
plague there may be no obvious signs.
Prevention and Treatment
Hospitalization and strict isolation are the rule
for this serious but easily treatable disease.
Streptomycin and gentamycin
are highly effective but other effective antibiotics include tetracycline,
fluoroquinalones and chloramphenicol. For most recent CDC
recommendations go
here.
An effective formalin-killed vaccine is available but is
recommended only for people at a high risk. The disease is internationally quarantinable and reporting of cases is mandatory. Control of urban plague is
based upon flea and rodent control.
Summary of types and symptoms of plague
Bubonic plague
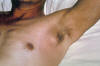 |
An
axillary bubo and edema exhibited by a plague patient
CDC/Margaret Parsons, Dr.
Karl F. Meyer |
This usually occurs as a result of a bite by an infected flea.
The lymph nodes closest to the bite become swollen as the bacteria
proliferate. They can then spread to other parts of the body.
Symptoms of bubonic plague Sudden onset of
- Fever
- Headache
- Weakness
- Formation of buboes (swollen, tender lymph nodes). See
below
Pneumonic plague
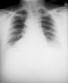
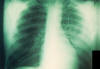 |
Anteroposterior chest x-ray of a
plague patient revealing bilateral infection,
greater on the patient's left side, which was
diagnosed as a case of pneumonic plague, caused
by the bacterium, Yersinia pestis.
CDC/ Dr. Jack
Poland |
This is usually the result of inhaling aerosols from an
infected patient or animal. It can also develop as the
result of spread of bacteria to the lungs in other forms of
plague that remain untreated. It is the most serious form of
plague as it can easily be spread.
Symptoms of pneumonic plague
- Fever
- Headache
- Weakness
- Pneumonia (chest pain, cough with sometimes
watery or bloody mucous, shortness of breath)
Septicemic plague
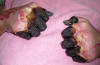 |
A 59 year-old man’s hands
who had been infected by the plague
bacterium, Yersinia pestis, after
having come into contact with both an
infected cat, and a dead mouse in his
neighborhood. The gangrenous condition
of the fingers had turned the dead
digits black, and mummified.
Two days after the
exposure the patient developed fever and
myalgias, and by the following day he
had developed a left axillary bubo.
Seven days after the initial exposure he
became critically ill and was admitted
to the hospital with multiple organ
failure. Initial blood cultures were
positive for double-curved,
Gram-negative Y. pestis rods.
The patient was
treated with gentamicin and survived,
but necrosis of the hands and feet
developed during hospitalization. He
subsequently required amputation of the
hands and feet.
CDC PHIL |

|
Swollen lymph glands (buboes) caused by plague bacteria in bubonic plague CDC |
This comes from flea bits or contact with an
infected animal. It may the initial disease or the
consequence of untreated bubonic plague.
Symptoms of septicemic plague
- Fever
- Chills
- Weakness
- Abdominal pain
- Shock
- Bleeding in skin and organs
- Skin, particularly at extremities,
becomes black and necrotic
|
|
|
 Male Xenopsylla cheopsis (oriental rat flea) engorged with blood CDC Male Xenopsylla cheopsis (oriental rat flea) engorged with blood CDC
 Wayson stain of
Yersinia pestis. Note the characteristic safety pin
appearance of bacteria CDC Wayson stain of
Yersinia pestis. Note the characteristic safety pin
appearance of bacteria CDC
 Symptoms in this plague patient include an inguinal bubo CDC
Symptoms in this plague patient include an inguinal bubo CDC
 Histopathology of spleen in fatal human plague - Necrosis and Yersinia
pestis. CDC/Dr. Marshall Fox
Histopathology of spleen in fatal human plague - Necrosis and Yersinia
pestis. CDC/Dr. Marshall Fox
 Histopathology of lymph node in fatal human plague - Medullary necrosis with fluid and Yersinia
pestis. CDC/Dr. Marshall Fox
Histopathology of lymph node in fatal human plague - Medullary necrosis with fluid and Yersinia
pestis. CDC/Dr. Marshall Fox
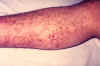 Capillary fragility is one of the manifestations of a plague infection, evident here on the leg of an infected patient.
CDC
Capillary fragility is one of the manifestations of a plague infection, evident here on the leg of an infected patient.
CDC
 Gangrene is one of the manifestations of plague, and is the origin of the term "Black Death" given to plague throughout the ages
CDC
Gangrene is one of the manifestations of plague, and is the origin of the term "Black Death" given to plague throughout the ages
CDC
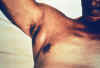 Plague patient displying a swollen axillary lymph node CDC
Plague patient displying a swollen axillary lymph node CDC
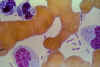 Dark stained bipolar ends of Yersinia pestis can clearly be seen in
this Wright's stain of blood from a plague victim
Dark stained bipolar ends of Yersinia pestis can clearly be seen in
this Wright's stain of blood from a plague victim
 Yersinia
pestis grows
well on most standard laboratory media, after 48-72 hours, grey-white to
slightly yellow opaque raised, irregular “fried egg” morphology;
alternatively colonies may have a “hammered copper” shiny
surface. CDC
Yersinia
pestis grows
well on most standard laboratory media, after 48-72 hours, grey-white to
slightly yellow opaque raised, irregular “fried egg” morphology;
alternatively colonies may have a “hammered copper” shiny
surface. CDC
|
|
|
|
|
 Robert Koch's original micrographs of
anthrax bacillus DOD Anthrax Program
Robert Koch's original micrographs of
anthrax bacillus DOD Anthrax Program
 Gram stain of anthrax DOD Anthrax Program
Gram stain of anthrax DOD Anthrax Program
|
ANTHRAX
Morphology and physiology
Bacillus anthracis is the causative agent of anthrax. It is a
Gram-positive, aerobic, spore-forming large bacillus. Spores are formed in
culture, in the soil, and in the tissues and exudates of dead animals, but not
in the blood or tissues of living animals. Spores remain viable in soil for
decades.
Epidemiology, transmission and
symptoms
Anthrax is a major disease threat
to herbivorous animals (cattle, sheep, and to a lesser extent horses, hogs, and
goats). People become infected by the cutaneous route (direct contact with
diseased animals, industrial work with hides, wool, brushes, or bone meal), by
inhalation (Woolsorter's disease), or by ingestion (meat from diseased
animals). It is not contagious.
|
|
 Anthrax due to Bacillus anthracis
(blood smear) ©
Bristol Biomedical Image Archive, University of Bristol. Used with
permission
Anthrax due to Bacillus anthracis
(blood smear) ©
Bristol Biomedical Image Archive, University of Bristol. Used with
permission

Under a very high magnification of 31,207X, this scanning
electron micrograph (SEM) depicted spores from the Sterne strain of
Bacillus anthracis bacteria CDC/ Laura Rose/Janice Haney Carr
 Cutaneous Anthrax DOD Anthrax Program
Cutaneous Anthrax DOD Anthrax Program
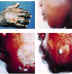 Cutaneous anthrax CDC Cutaneous anthrax CDC |
accounts
for more than 95% of human cases. Spores enter through small break in skin,
germinate into vegetative cells which rapidly proliferate at the portal of
entry. Within a few days, a small papule emerges that becomes vesicular. The
latter is filled with blue-black edema fluid. Rupture of this lesion will reveal
a black eschar
at the base surrounded by a zone of
induration.
This lesion is called a malignant pustule; however, no pus or pain are
manifested. The lesion is classically found on the hands, forearms or head. The
invasion of the bloodstream will lead to systemic dissemination of bacteria.
Pulmonary anthrax
results form inhalation of B. anthracis spores which are
phagocytized
by
the alveolar macrophages where they germinate and replicate. The injured host
cell and organisms infect the hilar lymph node where marked hemorrhagic
necrosis may occur. The patient may manifest fever, malaise, myalgia, and a
non-productive cough. Once in the hilar lymph node, infection may spread into the
blood stream. Respiratory distress and cyanosis are manifestations of toxemia.
Death results within 24 hours. This form of anthrax is of significance in biological warfare.
results from ingestion of meat-derived from an infected animal and leads to
bacterial proliferation within the gastrointestinal tract, invasion of the
epithelium and
ulceration of the mucosa. The invasion spreads to the mesenteric lymph nodes and
then to the bloodstream. Initially, there is vomiting and diarrhea followed by blood in
the feces. Invasion of the bloodstream is associated with profound prostration,
shock and death. Because of strict control measures, this form of anthrax is
not seen in the U.S. Without treatment of gastrointestinal anthrax, the majority
of patients die but with antibiotic treatment, 60% or more
survive.
Injection anthrax is are and has not been
seen in North America but has occurred in heroin users in
northern Europe. Symptoms are similar to cutaneous anthrax but
the infection may be deeper at the site of needle entry. The
bacteria can spread more rapidly from site of infection to other
parts of the body than is the case with cutaneous anthrax.
Meningeal Anthrax. All forms of anthrax
above can progress to meningeal encephalitis with deep brain
hemorrhagic lesions and infection of the cerebro-spinal fluid.
It is almost always fatal.
Summary of types and symptoms of anthrax
Cutaneous anthrax
Symptoms
-
Blisters near site of infection
-
After the blister, a painless ulcer appears with
a black center with swelling. This is most often on the face,
neck, arms or hands
 |
Cutaneous anthrax lesion on the
volar surface of the right forearm CDC |

|
Development of cutaneous anthrax CDC |
Pulmonary anthrax
Symptoms
Gastrointestinal anthrax
Symptoms
-
Fever and chills
-
Headache
-
Neck swelling
-
Red face and eyes
-
Sore throat, hoarseness and pain swallowing
-
Nausea and bloody vomit
-
Stomach cramps and stomach swelling
-
Diarrhea,
sometimes bloody
-
Faintness
-
Malaise
Injection anthrax
Symptoms
-
Fever and chills
-
Small blisters at site of drug injection
-
A painless ulcer with a black center with
swelling at site of injection after blisters appear
-
Deep abscess at injection site
|
|
CASE REPORT
Inhalation Anthrax Case
in Pennsylvania - 2006 |
| |
 Anthrax skin lesion on face of man CDC Anthrax skin lesion on face of man CDC
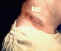 Anthrax skin lesion on neck of man CDC Anthrax skin lesion on neck of man CDC
 Marked hemorrhage in mucosa and submucosa with arteriolar
Marked hemorrhage in mucosa and submucosa with arteriolar
degeneration.
CDC/Dr. Marshall Fox
 Histopathology of liver in fatal human anthrax. Dilated sinusoids, neutrophil infiltrate.
CDC/Dr. Marshall Fox
Histopathology of liver in fatal human anthrax. Dilated sinusoids, neutrophil infiltrate.
CDC/Dr. Marshall Fox

53 year old
female, employed 10 years in the spinning department of a goat-hair
processing mill. Cutaneous anthrax lesion on right cheek; lesion as seen
on 4th day CDC
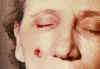
lesion as seen on 5th
day CDC
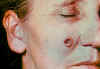
lesion as seen on
6th day CDC
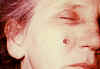
lesion as seen on 8th
day CDC
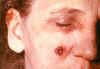
lesion as seen on 11th
day CDC
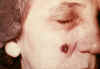
lesion as seen on 13th
day CDC
|
| |
| |
|
|
Pathogenesis
The virulence factors of B. anthracis include a number of exotoxins and
the capsule.
Exotoxin
A
plasmid-encoded, heat-labile, heterogeneous protein complex made up of 3
components:
-
Edema Factor (EF)
-
Lethal Factor (LF)
-
Protective Antigen (PA).
In vivo, these three factors act
synergistically (for toxic effects). The protective antigen binds to surface
receptors on eucaryotic cells and is subsequently cleaved by a cellular
protease. The larger C-terminal piece of PA remains bound to the receptor and
then binds either EF or LF, which enters the cell by
endocytosis. Edema Factor,
when inside the cells binds calmodulin-dependent and acts as adenylate cyclase.
Lethal factor's mechanism of action involves activation of macrophages and
production of cytokines which cause necrosis, fever, shock and death.
Individually, the three proteins have no known toxic activity. Antibodies to
protective antigens prevent PA binding to cells stop EF and LF entry.
Capsule
The capsule consists of a polypeptide of D-glutamic acid which is encoded by a
plasmid and is anti-phagocytic. It is not a good
immunogen and, even if any
antibodies produced, they are not protective against the disease.
Diagnosis
Clinical diagnosis of anthrax can be confirmed by direct examination or culture.
Fresh smears of vesicular fluid, fluid from under the eschar, blood, or spleen
or lymph node aspirates are stained with polychrome methylene blue and examined
for the characteristic blunt ended, blue-black rods with a pink capsule. In case
of a negative finding, the specimen can be cultured on blood agar plates.
Cultured organisms stain as Gram-positive long thin rods.
Prevention and Treatment
Antibiotics
Penicillin and the 4-quinolone,
Ciprofloxacin (Cipro), are the antibiotics of choice.
Anti-toxin
Antibody to the toxin complex is
neutralizing and protective. This may be used in combination with antibiotics.
There are two vaccines available. One is for use
for immunizing cattle and other herbivorous animals and the other for at-risk humans
(certain laboratory workers, people who handle animals (veterinarians)
and some military personnel.
|
|
|
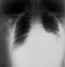 Mediastinal widening
and pleural effusion on chest X-ray in inhalational anthrax CDC
Mediastinal widening
and pleural effusion on chest X-ray in inhalational anthrax CDC
|
| |
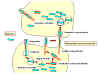 Infection of
macrophages or parenchymal cells by Listeria monocytogenes
Infection of
macrophages or parenchymal cells by Listeria monocytogenes
 Listeria monocytogenes - rod prokaryote that causes listeriosis, meningitis and food poisoning
©
Dennis Kunkel Microscopy, Inc.
Used with permission
Listeria monocytogenes - rod prokaryote that causes listeriosis, meningitis and food poisoning
©
Dennis Kunkel Microscopy, Inc.
Used with permission
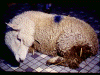
Live sheep: listeriosis
© Bristol Biomedical Image Archive, University of Bristol. Used with permission
 Listeria monocytogenes organisms in neutrophil
(blood smear) © Bristol Biomedical Image Archive,
University of Bristol. Used with permission
Listeria monocytogenes organisms in neutrophil
(blood smear) © Bristol Biomedical Image Archive,
University of Bristol. Used with permission
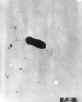 Electron micrograph of a flagellated Listeria monocytogenes bacterium, Magnified
41,250X
Electron micrograph of a flagellated Listeria monocytogenes bacterium, Magnified
41,250X
CDC/Dr. Balasubr Swaminathan; Peggy Hayes; Elizabeth White
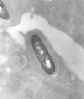 Electron micrograph of a Listeria bacterium in tissue.
Electron micrograph of a Listeria bacterium in tissue.
CDC/Dr. Balasubr Swaminathan; Peggy Hayes;
Elizabeth White |
LISTERIOSIS
Listeriosis is serious disease which is almost always caused by eating
contaminated food. It most affects newborn children, older and
immunocompromized people and pregnant women. There are about 1,600 cases of
listeriosis annually in the United States and about 260 deaths. In 2013, the
incidence of listeriosis was 2.6 cases per million.
Morphology and Physiology
L.monocytogenes is a facultative intracellular, Gram-positive
coccobacillus which often grows in short chains. It is different from other
Gram-positive organisms in that it contains a molecule chemically and
biologically similar to the classical lipopolysaccharide, the listerial LPS. The
organism forms beta hemolytic colonies on blood agar plates and blue-green
translucent colonies on colorless solid media. Upon infecting a cell
(macrophages and parenchymal cells), the organism escapes from the host vacuole
(or phagosome) and undergoes rapid division in the cytoplasm of the host cell
before becoming encapsulated by short actin filaments. These filaments
reorganize into a long tail extending from only one end of the bacterium. The
tail mediates movement of the organism through the cytoplasm to the surface of
the host cell. At the cell periphery, protrusions are formed that can then
penetrate neighboring cells and allow the bacterium to enter. Due to this mode
of cell-cell transmission, the organisms are never extracellular and exposed to
humoral antibacterial agents (e.g., complement, antibody). L.
monocytogenes is readily killed by activated macrophage.
Epidemiology and symptoms
Listeria monocytogenes is
a ubiquitous organism found in the soil, vegetation, water, and in the
gastrointestinal tract of animals. Exposure to the organism can lead to
asymptomatic miscarriage or disease in humans. At greatest risk for the
disease are the fetus, neonates, cancer patients and immuno-compromised
persons. In the United States, a number of recent outbreaks have been traced to
cheese, cole slaw (cabbage), milk, and meat. The organisms can grow at 4
degrees C which means that organism replication continues in refrigerated foods.
Laboratory isolation can employ a cold enrichment technique.
Listeriosis has been categorized in
two forms: 1) neonatal disease and 2) adult disease.
Neonatal Disease
Neonatal disease can occur in two forms:
-
Early onset disease,
acquired transplacetally in utero
In utero acquired infection (granulomatosus
infantiseptica) causes abscesses and granulomas in multiple organs and very
frequently results in abortion.
-
Late onset disease
acquired at birth or soon after birth.
Exposure on vaginal delivery results in
the late onset disease resulting in meningitis or meningo-encephalitis with
sepsis within 2 to 3 weeks.
Adult Disease
Infection in normal adults results in self-resolving flu-like symptoms
and/or mild gastrointestinal disturbance. Chills and fever are due to bacteremia.
In pregnant women, infection can lead to miscarriage, still birth or
premature birth with life-threatening
In immunosuppressed individuals
listeriosis can produce serious illness,
leading to meningitis. It is one of the leading causes of bacterial
meningitis in patients with cancer and in renal transplant recipients. In
the elderly, the early symptoms may go unnoticed and the infection may lead
to acute manifestations of sepsis (high fever, hypo-tension). A complication
of the bacteremia is endocarditis.
Pathogenesis
Listeriolysin O, a β-hemolysin,
is related to streptolysin, and pneumolysin and is produced by virulent strains. It disrupts the phagocytic vacuole and is
instrumental in cell-cell transmission of the organism. The toxin is oxygen
labile and immunogenic.
Diagnosis
Listeriosis is indicated when blood and CSF monocytosis is observed. The
organism can be isolated on most laboratory media.
Treatment and control
Penicillin (ampicillin) alone or in combination with gentamycin have been
effective. Immunity is cell-mediated.
|
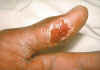 Thumb with skin ulcer of tularemia.
CDC/Emory U./Dr. Sellers
Thumb with skin ulcer of tularemia.
CDC/Emory U./Dr. Sellers
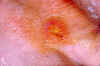 Tularemia lesion on the dorsal skin of right hand
CDC/Dr. Brachman
Tularemia lesion on the dorsal skin of right hand
CDC/Dr. Brachman
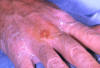
Tularemia on the hand CDC
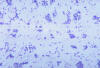 Francisella tularensis bacteria stained with methylene blue CDC/Dr.
P. B. Smith
Francisella tularensis bacteria stained with methylene blue CDC/Dr.
P. B. Smith
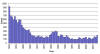 Reported cases of tularemia in the United States 1050-2010
CDC
Reported cases of tularemia in the United States 1050-2010
CDC
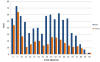 Average annual
incidence rate of tularemia by sex and age group - United States,
2001-2010 CDC
Average annual
incidence rate of tularemia by sex and age group - United States,
2001-2010 CDC
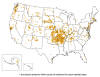
Reported tularemia cases in the United States - 2004 - 2013
CDC

Reported cases of tularemia by month in the United States
2001-2010 CDC
|
TULAREMIA
Morphology and physiology
Francisella tularensis is a small, Gram-negative,
non-motile, encapsulated,
pleomorphic coccobacillus (short rod). It is a facultative intracellular
parasite which grows poorly or not at all on most laboratory media and requires a
special glucose cysteine blood agar for isolation. Care must be taken in
handling the sample because of the low infectious dose.
Epidemiology and symptoms
Francisella tularensis is the causative agent of tularemia (a reportable
disease in the U.S.). Unlike plague, tularemia occurs routinely in all 50 of the
United States. Its primary reservoirs are rabbits, hares, rodents and ticks.
People most
commonly acquire tularemia via insect bites (ticks primarily, but also deer
flies, mites, blackflies, or mosquitoes) or by handling infected animal tissues.
Human disease (rabbit or deer fly fever) is characterized by a focal ulcer at
the site of entry of the organisms and enlargement of the regional lymph nodes.
In 2013, there were 203 cases of tularemia in the United
States (incidence: 0.6 cases per million population). Because the disease is
spread by ticks and flies, it is most common in the summer months.
As few as10 - 50 bacilli will cause
disease in humans if inhaled or introduced intradermally, whereas a very large
inoculum (~108 organisms) is required for the oral route of
infection. Incubation period is 3 - 10 days. A small skin papule usually develops
at the site of entry. Ulceration occurs together with fever, chills, malaise,
fatigue, and usually lymphadenopathy. Bacteremia usually occurs and the bacilli
then grow intracellularly in the reticuloendothelial system. Dissemination of
the organisms through the bloodstream permits focal lesions to develop in
numerous organs. The patient will normally exhibit one of several clinical
syndromes and the infection can be life-threatening, although most cases are
mild.
Forms of tularemia
Ulceroglandular tularemia
This form is most common (70 - 85% of cases) in which a
painful ulcerating papule, which has a necrotic center and raised periphery,
develops at the site of infection (usually from a tick or fly bite). The patient
experiences a fever (which can be as high as 104 degrees F) together
with lymph node swelling particularly in the arm pit and groin.
Glandular tularemia
This is acquired in the same way as ulceroglandular tularemia; however,
there is lymphadenopathy but no ulcer.
Oculoglandular
tularemia
This is often acquired when a person handling infected meat rubs the
bacteria into the eye. There is inflammation of the eye and swelling of
regional lymph glands.
Pneumonic tularemia
This is a very serious disease that
comes from breathing in the bacteria in dust or aerosols. It results in
difficulty breathing, chest pain and cough. It can also result from not
treating other forms of tularemia resulting is dissemination to the
lungs.
Oropharyngeal
tularemia
In this form, the disease is acquired by eating or drinking contaminated
food or drink. Patients experience pharyngotoncillitis
with lymphadenopathy accompanied by mouth ulcers. This is a rare form of
tularemia.
Pathogenesis
The capsule of the organism renders it resistant to phagocytosis.
Intracellularly, the organisms resist killing by phagocytes and multiply. Most
of the symptoms are due to cell-mediate hypersensitivity.
Diagnosis
F. tularensis is difficult to visualize in direct smears. The organism
can be isolated from specimens of sputum, or lymph node aspirates inoculated on
chocolate blood agar. Blood cultures are often negative. The organism grows very
slowly and hence must be incubated for several days. The identity of the
organism is confirmed with specific antisera.
Prevention and treatment
Streptomycin is the drug of choice for all forms of tularemia. Untreated, cases
have a fatality rate of 5 - 15%. A live attenuated organism vaccine is available
but its use is restricted to those persons who are at risk. Immunity appears to
be cell mediated. One must avoid handling infected animals, watch out for ticks
and utilize clean water supplies.
|
|
WEB RESOURCES
CDC-MMWR
Tularemia --- United States, 1990--2000
|
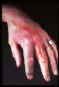 Erysipeloid- is an acute but very slowly evolving skin infection caused by
gram-positive bacillus (Erysipelothrix rhusiopathiae).
©
Mount
Allison Science Image Collection
Erysipeloid- is an acute but very slowly evolving skin infection caused by
gram-positive bacillus (Erysipelothrix rhusiopathiae).
©
Mount
Allison Science Image Collection |
ERYSIPELOID
This is an occupational disease of
butchers, meat processors, farmers, poultry workers, fish handlers: swine and
fish handlers are particularly at risk. The causative agent, Erysipelothrix
rhusiopathiae, is a Gram-positive anaerobic rod which infects through skin
abrasion while handling contaminated animal products or soil. Generally, the
organism produces an inflammatory skin lesion, on fingers or hand, which is
violaceus and has a raised edge. It spreads peripherally, as the discoloration
in the central area fades. The painful lesion is
pruritic and causes a burning
or throbbing sensation. It lacks suppuration and thus is distinguishable
from staphylococcal erysipelas. Diffuse cutaneous infection and septicemia are
rare. The organism can be cultured easily on most laboratory media. It is easily
treatable with penicillin.
|
|
|
 Return to the Bacteriology Section
of Microbiology and Immunology On-line Return to the Bacteriology Section
of Microbiology and Immunology On-line
This page last changed on
Sunday, February 14, 2016
Page maintained by
Richard Hunt
|

 Brucella abortus - Gram-negative, coccobacillus prokaryote; causes bovine spontaneous abortion due to its rapid growth in the presence of
erythritol (produced in the plancenta). SEM x 29,650
©
Dennis Kunkel Microscopy, Inc.
Used with permission
Brucella abortus - Gram-negative, coccobacillus prokaryote; causes bovine spontaneous abortion due to its rapid growth in the presence of
erythritol (produced in the plancenta). SEM x 29,650
©
Dennis Kunkel Microscopy, Inc.
Used with permission
 Plague suits. The beak was filled with sweet smelling oils and vinegar
to counteract the smell of plague victims. Although physicians in the 1300's
did not know the cause of plague, the suits may have been effective to some
degree in that they kept fleas off and the shiny beak may have posed an
obstacle to the entry of fleas. Left From:
Bubonic Plague by Ely Janis Right:
©
Bristol Biomedical Image Archive, University of Bristol. Used with
permission
Plague suits. The beak was filled with sweet smelling oils and vinegar
to counteract the smell of plague victims. Although physicians in the 1300's
did not know the cause of plague, the suits may have been effective to some
degree in that they kept fleas off and the shiny beak may have posed an
obstacle to the entry of fleas. Left From:
Bubonic Plague by Ely Janis Right:
©
Bristol Biomedical Image Archive, University of Bristol. Used with
permission
 Male Xenopsylla cheopsis (oriental rat flea) engorged with blood CDC
Male Xenopsylla cheopsis (oriental rat flea) engorged with blood CDC
 Histopathology of spleen in fatal human plague - Necrosis and Yersinia
pestis. CDC/Dr. Marshall Fox
Histopathology of spleen in fatal human plague - Necrosis and Yersinia
pestis. CDC/Dr. Marshall Fox
 Capillary fragility is one of the manifestations of a plague infection, evident here on the leg of an infected patient.
CDC
Capillary fragility is one of the manifestations of a plague infection, evident here on the leg of an infected patient.
CDC Plague patient displying a swollen axillary lymph node CDC
Plague patient displying a swollen axillary lymph node CDC Yersinia
pestis grows
well on most standard laboratory media, after 48-72 hours, grey-white to
slightly yellow opaque raised, irregular “fried egg” morphology;
alternatively colonies may have a “hammered copper” shiny
surface. CDC
Yersinia
pestis grows
well on most standard laboratory media, after 48-72 hours, grey-white to
slightly yellow opaque raised, irregular “fried egg” morphology;
alternatively colonies may have a “hammered copper” shiny
surface. CDC Anthrax skin lesion on face of man CDC
Anthrax skin lesion on face of man CDC

 Mediastinal widening
and pleural effusion on chest X-ray in inhalational anthrax CDC
Mediastinal widening
and pleural effusion on chest X-ray in inhalational anthrax CDC
 Infection of
macrophages or parenchymal cells by Listeria monocytogenes
Infection of
macrophages or parenchymal cells by Listeria monocytogenes
 Thumb with skin ulcer of tularemia.
CDC/Emory U./Dr. Sellers
Thumb with skin ulcer of tularemia.
CDC/Emory U./Dr. Sellers
 Erysipeloid- is an acute but very slowly evolving skin infection caused by
gram-positive bacillus (Erysipelothrix rhusiopathiae).
©
Mount
Allison Science Image Collection
Erysipeloid- is an acute but very slowly evolving skin infection caused by
gram-positive bacillus (Erysipelothrix rhusiopathiae).
©
Mount
Allison Science Image Collection