| x | x | ||||
 |
|||||
| INFECTIOUS DISEASE | BACTERIOLOGY | IMMUNOLOGY | MYCOLOGY | PARASITOLOGY | VIROLOGY |
|
|
|
||||
| TURKISH | |||||
| ALBANIAN | |||||
|
Let us know what you think |
|||||
| SEARCH | |||||
|
|
|||||
|
|
|||||
|
|
BLASTOMYCOSIS (Blastomyces
dermatitidis)
Most of the systemic
fungi have a specific niche in nature where they are commonly found. Blastomyces
dermatitidis survives in soil that contains organic debris (rotting wood, animal
droppings, plant material) and infects people collecting firewood, tearing down
old buildings or engaged in other outdoor activities which disrupt the soil. In
addition to an ecological niche, most fungi that cause systemic infections have
a limited geographic distribution where they occur most frequently.
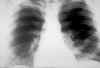
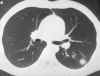
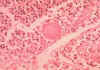
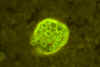
Blastomycosis (figures 1- 4)occurs in eastern North America (figure 5) and Africa. The vast majority of
patients with blastomycosis in South Carolina are infected in the northern
part of the state, above the Fall Line (Augusta, GA, Aiken, Columbia, Cheraw,
Raleigh, NC). |
||||
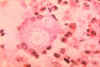
 Figure 1
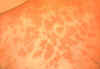
Figure 1Skin lesions of blastomycosis. |
|||||
|
|||||
 Figure 5
Figure 5Map of eastern United States and Canada showing distribution of reported cases of blastomycosis. CDC
|
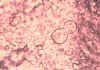
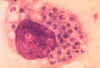
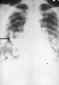
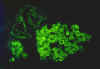
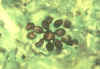
Blastomycosis is a chronic granulomatous disease which means that it progresses slowly. Although the pulmonary and skin (figure 1) involvement is the most common, B. dermatitidis frequently affects bone, prostate and other organs. More frequently blastomycosis presents as a cutaneous or a respiratory disease. The cutaneous lesions may be primary (usually self-limiting) or secondary (a manifestation of systemic disease). The patient who presents with a complaint of respiratory symptoms will frequently remark about loss of appetite, loss of weight, fever, productive cough, and night sweats. While these symptoms resemble those of TB, it is not this disease. The X-ray (figure 6) shows obvious pulmonary disease. To make the specific diagnosis, the physician must be aware of blastomycosis. Sputum sent to the lab for "culture" will not grow the organism. The lab must be alerted to look for fungal organisms or to look specifically for blastomyces. Some patients have a sub-clinical or "flu-like" response to infection. B. dermatitidis can frequently be demonstrated in a KOH preparation of pus from a skin lesion. A typical cutaneous lesion shows central healing with microabscesses at the periphery. A pus specimen may be obtained by nicking the top of a microabscess with a scalpel, obtaining the purulent material and making the diagnosis in 5 min. by microscopic examination with KOH. This organism has a characteristic appearance of a double contoured wall with a single bud on a wide base (figures 2 - 4). There are no specific virulence factors for B. dermatitidis. Laboratory specimens depend on the manifestation of the disease: If there are skin lesions, send skin scrapings or pus. If there is pulmonary involvement, send sputum. Other specimens include biopsy material and urine. Occasionally, the organism can be isolated from urine as it often infects the prostate.
|
||||
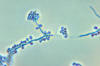
 Figure 7
Figure 7Spores of Histoplasma Dr Arthur DiSalvo |
HISTOPLASMOSIS (Histoplasma
capsulatum)
Histoplasmosis is a systemic disease, mostly of the reticuloendothelial system, manifesting itself in the bone marrow, lungs (figure 12-17), liver, and the spleen. In fact, hepatosplenomegaly is the primary sign in children, while in adults, histoplasmosis more commonly appears as pulmonary disease. This is one of the most common fungal infections, occurring frequently in South Carolina, particularly the northwestern portion of the state. The ecological niche of H. capsulatum is in blackbird roosts, chicken houses and bat guano. Typically, a patient will have spread chicken manure around his garden and 3 weeks later will develop pulmonary infection. There have been several outbreaks in South Carolina where workers have cleared canebrakes which served as blackbird roosts with bulldozers. All who were exposed, workers and bystanders, contracted histoplasmosis. Histoplasmosis is a significant occupational disease in bat caves in Mexico when workers harvest the guano for fertilizer. In the endemic area the majority of patients who develop histoplasmosis (95%) are asymptomatic. The diagnosis is made from their history, serologic testing or skin test. In the patients who are clinically ill, histoplasmosis generally occurs in one of three forms: acute pulmonary, chronic pulmonary or disseminated. There is generally complete recovery from the acute pulmonary form (another "flu-like" illness). However, if untreated, the disseminated form of disease is usually fatal. Patients will first notice shortness of breath and a cough which becomes productive. The sputum may be purulent or bloody. Patients will become anorexic and lose weight. They have night sweats. This again sounds like tuberculosis, and the lung x- ray also looks like tuberculosis, but today radiologists can distinguish between these diseases on the chest film (histoplasmosis usually appears as bilateral interstitial infiltrates). Histoplasmosis is prevalent primarily in the eastern U.S. In S.C., a histoplasmin skin test survey of lifetime, one county residents, white males, 17 to 21 years old, was performed on Navy recruits. The greatest number of positive skin tests appeared in the northwestern part of the state. A similar study of medical students conducted at Medical University of South Carolina, about 25 years ago, bore the same distribution.The skin test is NOT used for diagnostic purposes, because it interferes with serological tests. Skin tests are used for epidemiological surveys. Clinical specimens sent to the lab depend on the presentation of the disease: Sputum or Bronchial alveolar lavage, if it is pulmonary disease, or biopsy material from the diseased organ. Bone marrow is an excellent source of the fungus, which tends to grow in the reticulo-endothelial system. Peripheral blood is also a source of visualizing the organism histologically. The yeast (figures 7-11) is usually found in monocytes or in PMN's. Many times an astute medical technologist performing a white blood cell count will be the first one to make the diagnosis of histoplasmosis. In peripheral blood, H. capsulatum appears as a small yeast about 5-6 microns in diameter. (Blastomyces is 12 to 15 microns). Gastric washings are also a source of H. capsulatum as people with pulmonary disease produce sputum and frequently swallow their sputum.
|
||||
|
|
|||||
|
|||||
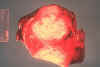 Figure 12.
Figure 12.Gross pathology specimen of lung showing cut surface of fibrocaseous nodule due to Histoplasma capsulatum. ASCP Atlas of Clinical Mycology II / CDC |
|
||||
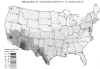 Figure 18
Figure 18Map of United States showing geographic variation in the prevalence of coccidioidin sensitivity in young adults CDC
|
Coccidioidomycosis is primarily a pulmonary disease. About 60 % of the infections in the endemic area are asymptomatic. About 25 % suffer a "flu-like" illness and recover without therapy. This disease exhibits the typical symptoms of a pulmonary fungal disease: anorexia, weight loss, cough, hemoptysis, and resembles TB. CNS infection with C. immitis is more common while it is less frequent with the other fungal diseases. The ecological niche of C. immitis is the Sonoran desert, which includes the deserts of the Southwest (California, Arizona, New Mexico, Nevada, Utah and Texas) and northern Mexico (figure 18). It is also found in small foci in Central and South America. Desert soil, pottery,
archaeological middens, cotton, and rodent burrows all harbor C. immitis.
C. immitis is a dimorphic fungus with 2 life cycles. The organism follows
the SAPROPHYTIC cycle in the soil and the PARASITIC cycle in man or animals. The
saprophytic cycle starts in the soil with spores (arthroconidia) that develop
into mycelium. The mycelium then matures and forms alternating spores within
itself. The arthroconidia are then released, and germinate back into mycelia
(figure 19).
The parasitic cycle involves the inhalation of the arthroconidia by animals
which then form spherules filled with endospores (figure 20). The ambient temperature and
availability of oxygen appear to govern the pathway The organism can be carried
by the wind and therefore spread hundreds of miles in storms so the distribution
is quite wide. In 1978, cases were seen in Sacramento 500 miles north of the
endemic area, from a dust storm in Southern California. The spores of the
organism are readily airborne. The cases that occur in South Carolina are
usually in patients who have visited an endemic area and brought back pottery,
or blankets purchase from a dusty roadside stand, or in Navy and Air Force
personnel who were exposed when they were stationed in the endemic area. The
disease manifests itself after they are transferred to a base in South Carolina.
A few interesting cases occurred in cotton mills in Burlington and Charlotte,
N.C. The cotton, grown in the desert of the Southwest, was contaminated with the
fungus spores and the mill workers inhaled the spores while handling the raw cotton and
developed coccidioidomycosis. |
||||
 Figure 20A
Figure 20ASmear of exudate showing spherules of Coccidioides immitis. Experimental infection of mouse with soil sample. CDC
|
Clinical Specimens
|
||||
|
|||||
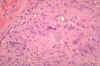 Figure 21
Figure 21Histopathology of coccidioidomycosis, retroperitoneal area. Coccidioides immitis fungi are visible within granuloma. CDC/Dr. Edwin P. Ewing, Jr. epe1@cdc.gov |
|
||||
 Figure 24
Figure 24Immune responses during coccidioidomycosis. Line graph showing immunologists' concept of the interplay between humoral and cell-mediated immune responses during coccidioidomycosis. TX State Chest Hosp/Dr. Rebecca A. Cox |
|
||||
 Figure 25
Figure 25Cervical lymphadenopathy Dr Arthur DiSalvo
|
PARACOCCIDIOIDOMYCOSIS (Paracoccidioides
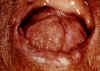
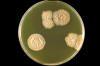
brasiliensis) This is a chronic granulomatous disease of mucous membranes, skin, and pulmonary system. This disease occurs from the middle of Mexico (North America) to Central and South America. Most cases are reported from Brazil. The ecological niche of this organism is probably the soil. A common triad of symptoms that are seen in Latin America is pulmonary lesions, edentulous mouth (figure 25 and 26), and cervical lymphadenopathy (figure 25). Prior to the recognition of this disease, patients in Latin America with paracoccidioidomycosis were often sent to TB sanitariums, just as patients with histoplasmosis were in the U.S. The organisms invade the mucous membranes of the mouth causing the teeth to fall out. White plaques are also found in the buccal mucosa, and this along with the triad are now used to clinically differentiate between TB and. This disease has a long latency period. 10-20 years may pass between infection and manifestation of the infection in the non-endemic areas of the world. Typically, a case of paracoccidioidomycosis seen in the U.S. occurs in someone who worked in South America for some period of time and then they return to the U.S. and years later, develop this disease. The patient does not realize the importance of this past history. Almost all diagnoses of fungal diseases depend upon careful questioning and a probing history. The clinical material which should be sent to the lab for examination is sputum, biopsy material, pus, and crust from the lesions. Examination of sputum or crust from one of the lesions with KOH reveals a yeast because this is a dimorphic fungus. In contrast to the other yeasts, particularly Blastomyces, Paracoccidioides has multiple buds, a thin cell wall, and a narrow base. At 25 degrees C, the colony is a dense, white mycelium (figure 28), not loose and cottony like the others. On Sabouraud's agar (figure 29), it takes 2-3 weeks to grow. When cultured at 37 degrees C, it is slow growing with a white-tan, thick colony. Microscopically, these yeasts appear as described above ranging in size from 5 to 15 microns.
|
||||
 Figure 28
Figure 28Lowenstein-Jensen slant culture of the fungus Paracoccidioides brasiliensis grown at 37°C. CDC/Dr. William Kaplan |
|
||||
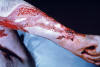
 Figure 33
Figure 33This patient presented with sporotrichosis affecting the skin of the thumb. CDC
|
SPOROTRICHOSIS (Sporothrix schenckii) Sporotrichosis is usually a chronic
infection of the cutaneous or subcutaneous tissue which tends to suppurate,
ulcerate and drain. In recent years, a pulmonary disease has been seen more
frequently. Occasionally, infection with S. schenckii may result in a
mycetoma. Sporotrichosis is caused by another dimorphic fungus. The infection is
also known as "rose growers disease." The ecologic niche for this
organism is rose thorns, sphagnum moss, timbers and soil. A study on the
occupational distribution of sporotrichosis showed that forest employees
accounted for 17% of the cases, gardeners and florists, 10%; and other
soil-related occupations another 16%. Sporotrichosis occurs worldwide. Every
aspect of this disease (clinical, pathology, mycology, ecology) was investigated
during an epidemic of 3,000 patients in a gold mine in South Africa during the
1940's. Patient history is very important in this disease also. It is often seen
in gardeners and begins with a thorn prick on the thumb (figure 33). A pustule develops and
ulcerates. It infects the lymphatic system and then the disease progresses up
the arm with ulceration (figure 39), abscess formation, break down of the abscess with large
amounts of pus followed by healing. Progression usually stops at the axilla.
Clinical material to be sent to the lab may be pus, biopsy material, or sputum
from pulmonary patients. The yeast form of this fungus in tissue or in culture,
can be round (6 - 8 um) or fusiform (figure 34). The fusiform shape is not the usual
form but if a cigar-shaped yeast (figure 34) is observed in tissue, it is usually diagnostic
of sporotrichosis. S. schenckii does not stain with the usual
histopathological stains. If sporotrichosis is suspected, the pathologist must
be informed so he can use special stains. Histologically asteroid bodies, a
tissue reaction (also known as Splendori reaction) may be seen around the yeast
cell. At 25 degrees C, this colony is white-cream and very membranous (figure 36-38), but as it ages for
2-3 weeks it becomes black and leathery (figure 36). Microscopically, the mycelium is
branching, septate and very delicate, 2-3 um in diameter (figure 35). The pyriform conidia,
2-4 um form a typical arrangement in groups at the end of a conidiophore
called "daisies" (figure 35). Serologic tests are not commercially
available. For the systemic form the drug of
choice is itraconazole or amphotericin B. |
||||
|
|
|||||
 Figure 37
Figure 37SABHI agar slant culture of the fungus Sporothrix schenckii grown at 37 degrees CDC/Dr. William Kaplan |
|
||||
|
|
|||||